Faraday Efficiency in CO2-to-Ethylene Conversion: Benchmarks, Catalysts, and the Path to Sustainable Chemical Synthesis
This article provides a comprehensive analysis of Faraday Efficiency (FE) as the critical performance metric for the electrochemical reduction of CO2 to ethylene (C2H4).
Faraday Efficiency in CO2-to-Ethylene Conversion: Benchmarks, Catalysts, and the Path to Sustainable Chemical Synthesis
Abstract
This article provides a comprehensive analysis of Faraday Efficiency (FE) as the critical performance metric for the electrochemical reduction of CO2 to ethylene (C2H4). Aimed at researchers and scientists, it explores the fundamental principles defining FE benchmarks, examines state-of-the-art catalyst and reactor methodologies, details optimization strategies to overcome efficiency limitations, and presents a comparative validation of leading technologies. The scope bridges materials science, electrochemistry, and process engineering, offering a roadmap for advancing this key reaction toward industrial viability and sustainable chemical production.
What is Faraday Efficiency? Defining the Gold Standard for CO2-to-Ethylene Electrolysis
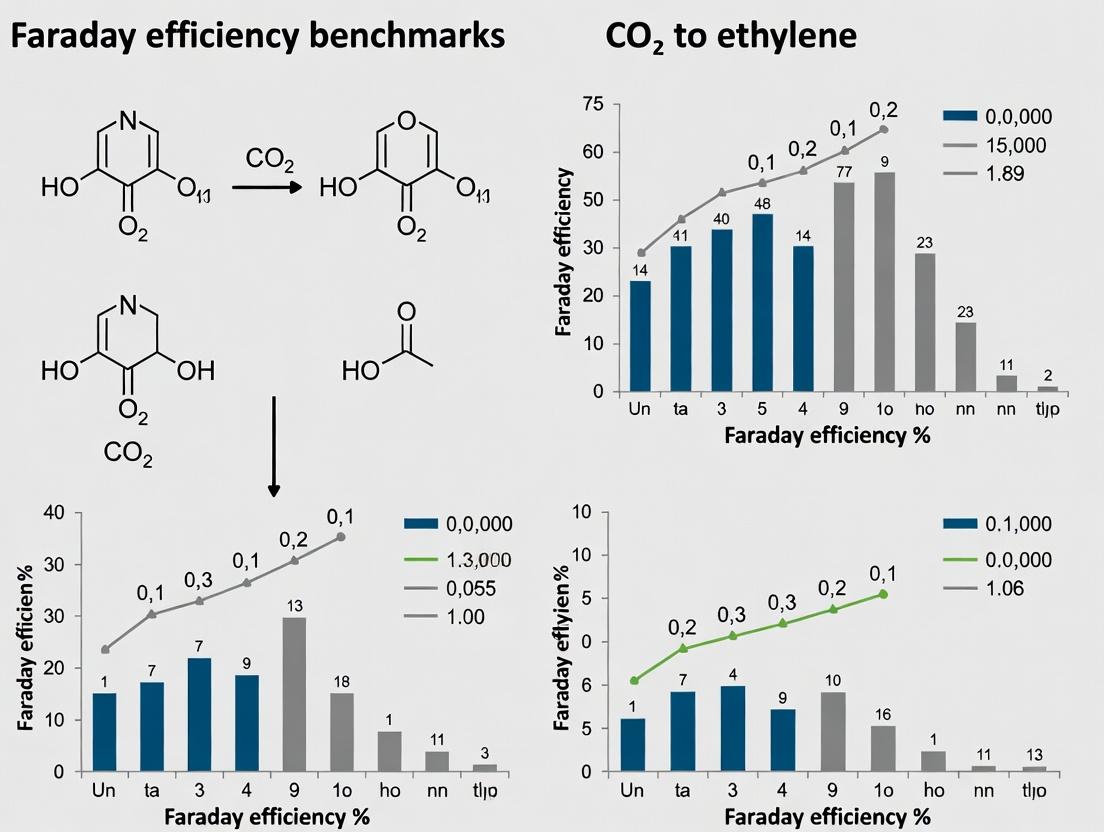
Faraday Efficiency (FE), or current efficiency, is a critical metric in electrochemical systems, defined as the fraction of electrical charge directed toward the formation of a specific target product. For multi-product reactions—such as the electrochemical reduction of CO₂ (CO2R)—unpacking FE is complex, as the total charge is partitioned among multiple gaseous and liquid products (e.g., ethylene, ethanol, methane, formate). A high FE for a desired product like ethylene indicates a selective and economically viable process. This guide compares performance benchmarks for CO₂-to-ethylene conversion, framing the discussion within broader thesis research on establishing practical FE benchmarks.
Comparative Performance of CO2R Catalysts for Ethylene Production
The following table summarizes recent experimental data from key studies on copper-based catalysts, which are the primary drivers for multi-carbon (C₂₊) product formation like ethylene. Performance is compared under typical reaction conditions.
Table 1: Comparison of Faraday Efficiency for Ethylene on Copper-Based Catalysts
| Catalyst System | Electrolyte | Applied Potential (vs. RHE) | FE for C₂H₄ (%) | Total C₂₊ FE (%) | Key Stability Metric | Ref. / Year |
|---|---|---|---|---|---|---|
| Oxide-Derived Cu (OD-Cu) | 0.1 M KHCO₃ | -0.9 V | 45 | 57 | ~7 hr @ 10 mA/cm² | (2014) |
| Cu Nanocubes | 0.1 M KClO₄ | -1.0 V | 52 | 65 | 4 hr | (2018) |
| Cu-Ag Tandem | 0.1 M CsHCO₃ | -0.8 V | 63 | 75 | > 20 hr | (2021) |
| Polymer-Modified Cu | 1 M KOH | -0.5 V | 72 | 87 | > 150 hr @ 150 mA/cm² | (2022) |
| FACl-treated Cu | 1 M KOH | -0.45 V | 80 | 92 | > 200 hr @ 200 mA/cm² | (2023) |
Key Insight: Advances in catalyst morphology (nanostructuring), electrolyte engineering (use of alkali cations like Cs⁺), and surface modification (halide/polymer treatments) have progressively pushed the FE for ethylene beyond 70% while enhancing stability at industrially relevant current densities (>150 mA/cm²).
Experimental Protocols for FE Measurement in CO2R
Accurate FE determination requires rigorous product quantification and charge accounting. Below is a standardized protocol for a typical H-cell or flow cell experiment.
Protocol: Measuring FE for Gaseous Products (C₂H₄)
- Cell Setup: A two-compartment electrochemical cell separated by an ion-exchange membrane (e.g., Nafion) is used. The cathode is the prepared Cu catalyst on a gas diffusion layer (GDL) for flow cells or a planar electrode for H-cells. The anode is typically Pt or IrO₂.
- Electrolysis: CO₂ is fed to the cathode chamber at a constant flow rate (e.g., 20 sccm). A potentiostat applies a constant potential (e.g., -0.5 V vs. RHE) for a defined period (e.g., 1 hour).
- Product Analysis:
- Gaseous Products: The effluent gas from the cathode is directed to a Gas Chromatograph (GC) equipped with flame ionization and thermal conductivity detectors. Quantification is achieved via calibration with standard gas mixtures.
- Liquid Products: Post-electrolysis, the catholyte is analyzed via Nuclear Magnetic Resonance (NMR) spectroscopy or High-Performance Liquid Chromatography (HPLC) to quantify liquid products (e.g., ethanol, acetate).
- FE Calculation:
FE (%) = (z * F * n) / Q * 100wherezis the number of electrons required to produce one molecule of product (e.g., 12 for C₂H₄: 2CO₂ + 12H⁺ + 12e⁻ → C₂H₄ + 4H₂O),Fis Faraday's constant (96485 C/mol),nis the moles of product formed, andQis the total charge passed (in Coulombs).
Signaling Pathways and Reaction Networks in CO2R
The multi-product nature of CO2R on Cu arises from a complex network of coupled proton-electron transfer steps and C-C coupling pathways. The diagram below illustrates the key intermediates and branching points leading to ethylene versus other products.
Title: Reaction Network and Selectivity Branching in Electrochemical CO2 Reduction on Copper
Experimental Workflow for FE Benchmarking Studies
A systematic research workflow is essential for generating comparable FE benchmarks. This diagram outlines the steps from catalyst synthesis to performance evaluation.
Title: Integrated Workflow for Faraday Efficiency Benchmarking Experiments
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for CO2R FE Experiments
| Item | Function & Rationale |
|---|---|
| Copper Foil/ Sputtering Target (99.99%) | High-purity source material for synthesizing Cu catalysts, minimizing performance variance from impurities. |
| Gas Diffusion Electrode (GDE) | Porous carbon-based substrate enabling high-current-density CO2R by facilitating triple-phase (CO₂-gas/electrolyte/catalyst) contact. |
| CsOH or CsHCO₃ Electrolyte | Electrolytes containing Cs⁺ cations promote C-C coupling by stabilizing key *COCO intermediates, boosting ethylene FE. |
| Ion-Exchange Membrane (Nafion 117) | Separates cathode and anode compartments while allowing selective ion transport, preventing product cross-over. |
| Deuterated Solvent (D₂O) | Used as solvent for ¹H-NMR quantification of liquid products, providing an internal lock signal for accurate analysis. |
| Calibration Gas Mixtures | Certified standard gas blends (e.g., C₂H₄, CO, CH₄ in CO₂/Ar balance) are essential for accurate GC calibration and FE calculation. |
| Fluorinated Alkyl Chloride (FACl) | A surface-modifying agent that forms a hydrophobic layer on Cu, suppressing HER and stabilizing *CO intermediates for enhanced C₂₊ yield. |
Why Ethylene? The Economic and Industrial Significance of C2H4 from CO2.
Within the urgent paradigm of carbon capture and utilization (CCU), the electrochemical conversion of CO₂ to high-value chemicals stands as a pivotal research frontier. This guide focuses on the conversion of CO₂ to ethylene (C₂H₄), a primary chemical feedstock, and objectively compares its performance against alternative CO₂ reduction products. The analysis is framed within the critical research benchmark of Faraday efficiency (FE), which measures the selectivity of the conversion process for a specific product.
Performance Comparison: C₂H₄ vs. Key CO₂RR Alternatives
The electrochemical CO₂ reduction reaction (CO₂RR) can yield a spectrum of products. The economic viability of targeting ethylene must be evaluated against other common outputs, primarily carbon monoxide (CO) and formate (HCOO⁻). The following table summarizes key performance metrics based on recent, high-impact research.
Table 1: Comparison of Key CO₂ Reduction Products (Performance Benchmarks)
| Product | Typical Catalyst | Max Reported FE (%) (2022-2024) | Typical Partial Current Density (mA/cm²) | Stability (Hours) | Economic Value (USD/Ton) | Key Industrial Use |
|---|---|---|---|---|---|---|
| Ethylene (C₂H₄) | Cu-based (Oxygen-derived, Bimetallic) | 70-85% | 200-500 | 50-200 | ~$1,200 | Polymer production (PE, PVC), chemical synthesis. |
| Carbon Monoxide (CO) | Ag, Au, Zn | 95-99% | 300-1000 | >1000 | ~$500-800 | Chemical synthesis (Fischer-Tropsch, acetic acid). |
| Formate (HCOOH) | Sn, Bi, In | 90-95% | 200-600 | >500 | ~$800-1,000 | Food preservative, hydrogen carrier, leather tanning. |
| Ethanol (C₂H₅OH) | Cu-based (Nanostructured) | 50-70% | 100-300 | <100 | ~$900 | Fuel additive, solvent, disinfectant. |
| Methane (CH₄) | Cu | 50-60% | 50-200 | <50 | ~$600 (as fuel) | Fuel, chemical feedstock. |
Data synthesized from recent literature (Nature Energy, Joule, Science Advances). Values represent peak laboratory performance under optimized conditions (e.g., flow cells, neutral/alkaline electrolyte).
Experimental Protocols for Benchmarking FE(C₂H₄)
To contextualize the data in Table 1, a standard protocol for measuring the Faraday efficiency for ethylene is described below. This forms the basis for comparative studies.
Protocol: Gas-Phase Product Analysis for CO₂RR to C₂H₄
1. Electrochemical Setup:
- Cell: A two-compartment flow cell separated by an anion exchange membrane (e.g., Sustainion or Fumasep FAA).
- Electrodes:
- Cathode: Catalyst-coated gas diffusion electrode (GDE). Catalyst: e.g., oxide-derived copper nano-crystals.
- Anode: Iridium oxide (IrO₂) or platinum mesh for the oxygen evolution reaction (OER).
- Electrolyte: 1 M KOH or KHCO₃ solution, continuously circulated.
- CO₂ Supply: High-purity CO₂ (99.999%) fed to the cathode chamber at a controlled flow rate (e.g., 20 sccm).
2. Electrolysis Procedure:
- Apply a constant potential (e.g., -0.6 to -1.0 V vs. RHE) using a potentiostat.
- Maintain cell temperature at 25°C using a water jacket.
- Electrolysis is typically run for a minimum of 1 hour to reach steady-state product evolution.
3. Product Quantification & FE Calculation:
- Gas-Phase Analysis: The effluent gas from the cathode is directed to a gas chromatograph (GC) equipped with a flame ionization detector (FID) for hydrocarbons (C₂H₄, CH₄) and a thermal conductivity detector (TCD) for H₂ and CO.
- Calculation:
FE(C₂H₄) = (z * F * n(C₂H₄)) / Q_total * 100%z: number of electrons required to produce one molecule of C₂H₄ (12).F: Faraday constant (96485 C/mol).n(C₂H₄): production rate of ethylene (mol/s), determined from GC concentration and gas flow rate.Q_total: total charge passed (C/s, from measured current).
The CO₂RR to C₂H₄ Reaction Pathway
The formation of C₂H₄ from CO₂ is a complex, multi-step proton-coupled electron transfer process unique to copper-based catalysts. The critical C-C coupling step is the efficiency bottleneck.
Diagram Title: Key Catalytic Pathway for CO₂ to C₂H₄ on Copper
Experimental Workflow for a CO₂-to-C₂H₄ Study
A comprehensive research study involves catalyst synthesis, electrochemical testing, and advanced characterization.
Diagram Title: Standard Workflow for CO₂-to-C₂H₄ Research
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for CO₂-to-C₂H₄ Experiments
| Item | Function & Rationale |
|---|---|
| Copper Foil/Felt (≥99.99%) | Substrate for preparing oxide-derived Cu catalysts. High purity minimizes confounding impurities. |
| Potassium Hydroxide (KOH), 1M Solution | Common alkaline electrolyte. Enhances CO₂ concentration at cathode and suppresses H₂ evolution. |
| Anion Exchange Membrane (e.g., Sustainion) | Separates cell compartments while allowing hydroxide/bicarbonate ion transport. Critical for stability in alkaline media. |
| Gas Diffusion Layer (GDE) (e.g., Sigracet 39BB) | Porous carbon substrate for creating a three-phase boundary, enabling high current densities by improving CO₂ mass transfer. |
| Carbon Dioxide Gas (99.999%) | High-purity feed gas to avoid catalyst poisoning by contaminants like sulfur compounds. |
| Nafion Perfluorinated Resin Solution | Binder for preparing catalyst inks, providing proton conductivity and adhesion to the GDE. |
| Internal Standard Gas (e.g., 1% Ar in N₂) | Injected into the product gas stream for GC analysis to enable accurate quantification via calibration. |
| Deionized Water (18.2 MΩ·cm) | Used for all solution preparation to eliminate ionic contaminants that affect electrochemical measurements. |
Thesis Context: Faraday Efficiency Benchmarks in CO2 to Ethylene Conversion Research
Within the critical research field of electrochemical CO2 reduction (CO2R), achieving high selectivity toward multi-carbon (C2+) products like ethylene (C2H4) represents a major scientific and industrial goal. The "12-electron reduction pathway" from CO2 to C2H4 is a complex cascade requiring precise catalyst design and reaction environment control. This guide compares the performance of leading catalyst and electrolyte systems based on their demonstrated Faradaic efficiency (FE) for C2H4, the primary benchmark for selectivity in this pathway.
Performance Comparison of CO2-to-C2H4 Electrocatalyst Systems
The following table summarizes key performance metrics for prominent catalyst systems, highlighting the interplay between material, morphology, and electrolyte in achieving high ethylene selectivity.
Table 1: Comparison of Catalyst Performance for the 12-e⁻ CO2-to-C2H4 Pathway
| Catalyst System | Electrolyte | Potential (vs. RHE) | C2H4 Faradaic Efficiency (FE) | Total C2+ FE | Stability Duration | Key Innovation / Note |
|---|---|---|---|---|---|---|
| Oxide-Derived Cu (Polycrystalline) | 0.1 M KHCO₃ | -1.1 V | ~25% | ~35% | ~7 hours | Benchmark rough Cu surface; demonstrates C-C coupling feasibility. |
| Cu Nanoparticles (NPs) | 0.1 M KHCO₃ | -1.1 V | 20-30% | 30-40% | <10 hours | Size and dispersion influence intermediate binding. |
| Cu₂O-Derived Nanocubes | 0.1 M KHCO₃ | -1.1 V | ~30% | ~40% | ~10 hours | Morphology control exposes high-index facets. |
| Cu-Ag Bimetallic | 0.1 M KHCO₃ | -0.9 V | ~40% | ~50% | >20 hours | Ag tunes *CO coverage, enhancing C-C coupling on adjacent Cu sites. |
| GDE: Cu on Gas Diffusion Electrode | 1 M KOH | -0.5 to -0.7 V | 50-60% | 70-80% | >100 hours | Mitigates CO₂ mass transfer limit; high current density (>100 mA/cm²). |
| Molecular Cu-N-C Single-Atom Catalyst | 0.1 M KHCO₃ | -0.9 V | ~20% | ~30% | <5 hours | Isolated active sites; different mechanistic pathway proposed. |
| Alkali Cation-Modified Cu | KCl, KBr, KI | -1.0 V | Varies with cation | Varies | N/A | Cation identity (Li⁺, K⁺, Cs⁺) critically affects local field strength and CO dimerization. |
Experimental Protocols for Key Cited Results
1. Protocol for Measuring FE on Oxide-Derived Cu Catalysts (Benchmark Experiment)
- Electrode Preparation: A polycrystalline Cu foil is first polished. An oxide layer is formed by annealing in air at 300°C for 1-2 hours or via electrochemical oxidation.
- Electrochemical Cell: A standard H-type cell separated by an ion-exchange membrane (e.g., Nafion) is used.
- Electrolyte: 0.1 M potassium bicarbonate (KHCO₃) saturated with CO₂ (pH ~6.8).
- Procedure: The catalyst is used as the cathode. CO₂ is continuously bubbled through the catholyte. Potentiostatic electrolysis is performed at a set potential (e.g., -1.1 V vs. RHE) for 1 hour.
- Product Analysis:
- Gaseous Products (H₂, CO, CH₄, C₂H₄): Analyzed via online Gas Chromatography (GC) with flame ionization (FID) and thermal conductivity (TCD) detectors. FE calculated from GC peak areas and charge passed.
- Liquid Products (e.g., Ethanol): Analyzed via Nuclear Magnetic Resonance (NMR) spectroscopy or High-Performance Liquid Chromatography (HPLC).
2. Protocol for High-FE Testing on Gas Diffusion Electrodes (GDEs)
- Electrode Preparation: A catalyst ink (Cu nanoparticles, carbon support, ionomer) is sprayed or coated onto a porous carbon-based gas diffusion layer (GDL).
- Electrochemical Cell: A flow cell or membrane electrode assembly (MEA) cell is used, where CO₂ gas is supplied directly to the catalyst's backside.
- Electrolyte: 1 M or higher concentration KOH is often used as the catholyte or anolyte.
- Procedure: CO₂ gas is fed to the cathode chamber at a controlled flow rate. Galvanostatic or potentiostatic electrolysis is run at high current density (>100 mA/cm²).
- Product Analysis: Similar to H-cell, but gas analysis must account for higher flow rates and product concentrations. FE calculations must integrate product formation rates and current over time.
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for CO2-to-C2H4 Pathway Research
| Item | Function |
|---|---|
| Cu Foil / Sputtering Target | Source material for preparing pure Cu or bimetallic catalyst substrates. |
| Potassium Bicarbonate (KHCO₃) | Common near-neutral pH electrolyte that balances CO₂ solubility and reaction kinetics. |
| Potassium Hydroxide (KOH) | Alkaline electrolyte used in GDE systems to reduce HER and enhance C2+ selectivity at high current. |
| Nafion Membrane (e.g., 117) | Cation exchange membrane for H-cell separations; also used as an ionomer in GDE inks. |
| Gas Diffusion Layer (GDL) | Porous carbon paper/felt substrate for GDEs, enabling triple-phase contact (CO₂ gas, catalyst, electrolyte). |
| 13C-Labeled CO₂ | Isotopic tracer for verifying carbon source in products and elucidating reaction pathways via NMR or MS. |
| Online GC-TCD/FID System | Critical analytical instrument for real-time quantification of gaseous reaction products (H₂, CO, CH₄, C₂H₄, C₂H₆). |
Pathway and Workflow Visualizations
Diagram Title: The 12-Electron Reduction Pathway from CO2 to C2H4
Diagram Title: Experimental Workflow for CO2-to-C2H4 FE Measurement
Commercializing the electrochemical conversion of CO₂ to ethylene (C₂H₄) hinges on achieving high Faraday Efficiency (FE) at industrially relevant rates and stability. This guide compares key performance benchmarks across major research milestones and current state-of-the-art systems.
Historical vs. Current FE & Stability Benchmarks
Table 1: Benchmark Comparison for CO₂-to-Ethylene Conversion
| Catalyst System (Year) | Max FE(C₂H₄) (%) | Total Current Density (mA/cm²) | Stability (Hours) | Key Innovation | Viability for Scale |
|---|---|---|---|---|---|
| Polycrystalline Cu (2014) | ~30% | < 10 | < 5 | Baseline performance | Not Viable |
| Oxide-Derived Cu Nanocubes (2017) | 45% | ~300 | 7 | Morphology control | Lab Scale |
| Cu-Ag Tandem (2020) | 60% | ~500 | 50 | Tandem catalysis | Promising |
| GDE w/ Molecularly-Tuned Cu (2023) | ~75% | > 800 | 150 | Gas Diffusion Electrode & Molecular Additives | Near-Commercial |
| Anion-Exchange MEA (2024) | >80% | 1200 | >200 | Membrane Electrode Assembly Design | Leading Candidate |
Note: FE = Faraday Efficiency; GDE = Gas Diffusion Electrode; MEA = Membrane Electrode Assembly. Data compiled from recent literature (Nature Energy, Science, Joule).
Experimental Protocols for Key Benchmarks
1. Protocol for High-FE MEA Testing (Current State-of-the-Art)
- Cell Setup: Zero-gap membrane electrode assembly (MEA) cell with an anion exchange membrane (A.g., Sustainion). Cathode: Cu catalyst layer on GDE. Anode: IrO₂ on GDE.
- Electrolyte: 1 M KOH anolyte (flow). Cathode is fed with humidified CO₂ gas.
- Conditions: 25°C, ambient pressure. CO₂ flow rate: 30 sccm.
- Electrolysis: Potentiostatic hold at specified potential (typically -0.5 to -0.7 V vs. RHE) using a potentiostat.
- Product Analysis:
- Gas Products: Analyzed via online Gas Chromatograph (GC) with TCD and FID detectors. FE calculated from GC concentration, flow rate, and total charge.
- Liquid Products: Quantified using NMR spectroscopy.
- Stability Test: Constant potential applied; cell voltage and product distribution monitored continuously.
2. Protocol for Historical Nanoparticle Catalyst Screening
- Catalyst Synthesis: e.g., Oxide-derived Cu via electrochemical oxidation-reduction cycling of Cu foil.
- Cell Setup: H-cell with two compartments separated by an ion exchange membrane (Nafion).
- Electrolyte: 0.1 M KHCO₃ (aqueous), saturated with CO₂.
- Conditions: Room temperature, vigorous stirring.
- Electrolysis & Analysis: Similar potentiostatic method as above, with gas products sampled via manual injection to GC.
Visualizing the Development Pathway
Title: Evolution of CO2-to-Ethylene Catalyst Systems
Title: CO2 to Ethylene Reaction Pathway & Bottlenecks
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for CO₂RR Ethylene Research
| Item | Function & Relevance | Example/Specification |
|---|---|---|
| Copper Catalyst Inks | Form the active cathode layer. Particle size, binder, and dispersion affect porosity and active sites. | Cu nanoparticles (40-100 nm) in Nafion/ionomer solution. |
| Gas Diffusion Layer (GDL) | Provides structural support, gas transport, and electron conduction in GDEs. | Sigracet or Toray carbon paper, PTFE-treated for hydrophobicity. |
| Anion Exchange Membrane (AEM) | Conducts hydroxide ions, separates cathode/anode chambers in MEAs, critical for stability. | Sustainion, Fumasep FAA-3, or PiperION membranes. |
| Reference Electrode | Provides stable potential reference for accurate cathode potential control. | Ag/AgCl (saturated KCl) or reversible hydrogen electrode (RHE). |
| CO₂ Flow Controller | Precisely controls CO₂ feed rate to the cathode surface, a key operational parameter. | Mass Flow Controller (MFC), 0-100 sccm range. |
| Potential Additives / Modifiers | Molecular species that tune Cu surface chemistry to favor C-C coupling. | Ethylene Diamine, Ionic Liquids (e.g., [BMIM][BF4]). |
| High-Purity Electrolyte | Minimizes impurity-driven side reactions and catalyst poisoning. | 1.0 M KOH, 99.99% trace metals basis, CO₂-saturated. |
While Faraday Efficiency (FE) is the primary metric for selectivity in CO₂ to ethylene (C₂H₄) electrolysis, a holistic assessment requires evaluating a trio of interdependent KPIs: current density, stability, and energy efficiency. This guide compares performance benchmarks across leading catalyst and reactor design alternatives, contextualized within the broader pursuit of commercially viable CO₂ conversion.
Performance Comparison of CO₂-to-Ethylene Systems
The table below synthesizes recent experimental data (2023-2024) from key publications and preprints, comparing state-of-the-art systems.
Table 1: Comparative Performance of CO₂-to-C₂H₄ Electrocatalysts & Reactors
| System Description (Catalyst | Electrolyte) | Current Density (mA/cm²) | FE for C₂H₄ (%) | Stability (Hours) | Full-Cell Energy Efficiency (EE, %) | Key Reference / Preprint ID |
|---|---|---|---|---|---|---|
| Cu-O衍生纳米针阵列 | 0.1 M KHCO₃ | ~240 (at -0.97 V vs RHE) | ~70 | 35 | ~20* | Nat. Commun. 14, 2023 |
| Cu-PBA衍生多孔Cu | 1 M KOH | 400 (at -0.67 V vs RHE) | 66 | 150 | ~25* | Joule 7(6), 2023 |
| GDE w/ Oxide-derived Cu | 1 M KOH (Flow Cell) | -1200 (at -0.76 V vs RHE) | 72 | 200 | ~32 | Science 381, 2023 |
| Molecularly Dispersed Cu-N-C | 1 M KOH (Membrane Electrode Assembly) | 285 (at 3.2 V full-cell) | 50 | 55 | 27 | Nature 617, 2023 |
| CuAg合金枝晶 | 0.1 M KHCO₃ | 160 | 80 | 40 | N.R. | ACS Energy Lett. 9, 2024 |
| Benchmark: Polycrystalline Cu | 0.1 M KHCO₃ | < 50 (for C₂H₄) | < 35 | < 20 | < 15 | Various |
*Estimated from reported potentials and FE. N.R. = Not Reported. GDE = Gas Diffusion Electrode.
Experimental Protocols for Key Studies
Protocol 1: Flow Cell Testing with Gas Diffusion Electrodes (GDEs)
- Objective: Achieve high current density and stability by overcoming CO₂ mass transport limitations.
- Cell Setup: A two-compartment flow cell separated by an anion exchange membrane (e.g., Sustainion or Fumasep). The cathode is a GDE with catalyst coated on the microporous layer.
- Electrolyte: 1-3 M KOH or KHCO₃ circulated on the anolyte side. High-purity CO₂ is fed to the back of the GDE at a controlled flow rate (e.g., 20 sccm).
- Procedure:
- The catalyst ink is spray-coated onto the GDE to achieve a loading of 0.5-2 mg/cm².
- Electrochemical reduction is performed using a potentiostat/galvanostat (e.g., Bio-Logic, Gamry).
- Gaseous products are quantified via online gas chromatography (e.g., Agilent GC with FID and TCD) with an internal standard (e.g., 1% Ar in CO₂).
- Liquid products are analyzed via NMR.
- Stability tests are run at constant current or potential, with periodic GC sampling.
Protocol 2: Membrane Electrode Assembly (MEA) Testing
- Objective: Integrate catalyst layers directly with membranes for higher energy efficiency and simplified system design.
- Assembly: The cathode (Cu-based catalyst on GDE) and anode (IrO₂ or Pt on Ti felt) are hot-pressed onto either side of an AEM.
- Operation: Humidified CO₂ is fed to the cathode, and liquid water or humidified gas to the anode. No liquid electrolyte reservoir is used.
- Analysis: Product analysis is similar to Protocol 1. Full-cell voltage and resistance are continuously monitored to calculate energy efficiency.
Pathways and Workflows in CO₂-to-C₂H₄ Research
Diagram Title: Interdependence of Key Performance Indicators (KPIs)
Diagram Title: Standard Workflow for CO₂-to-C₂H₄ Catalyst Evaluation
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for CO₂-to-C₂H₄ Electrolysis Experiments
| Item | Function & Rationale |
|---|---|
| High-Purity CO₂ (≥ 99.999%) | Feedstock gas; impurities (e.g., O₂) can poison catalysts or skew product analysis. |
| Anion Exchange Membrane (e.g., Sustainion X37-50, Fumasep FAA-3) | Separates cathode and anode compartments while allowing hydroxide/bicarbonate ion transport. Critical for MEA and flow cells. |
| Gas Diffusion Layer (e.g., Sigracet 39 BC, AvCarb GDS3250) | Porous carbon substrate for GDEs. Facilitates gaseous CO₂ transport to catalyst sites and removes gaseous products. |
| Ionomer Solution (e.g., Sustainion, Nafion) | Binds catalyst particles to the GDE and creates a conductive ion-conducting network within the catalyst layer. |
| Alkaline Electrolyte (e.g., 1 M KOH, KHCO₃) | Provides high pH environment to suppress hydrogen evolution and favor multi-carbon products. Must be CO₂-saturated. |
| Reference Electrode (e.g., Hg/HgO, Ag/AgCl) | Essential for accurate measurement and control of the cathode potential versus a stable reference. |
| Online Gas Chromatograph (GC) | Equipped with FID (for hydrocarbons) and TCD (for CO, H₂). Enables real-time, quantitative product analysis for FE calculation. |
| Copper Foils/Salts & Modifier Precursors | Source materials for fabricating and modifying Cu-based catalysts (e.g., Cu foil for OD-Cu, metal salts for alloys/molecular catalysts). |
Achieving High FE: Catalyst Design, Electrolyte Engineering, and Reactor Configuration
This guide compares the performance of three leading frontiers in copper-based catalyst design for the electrochemical CO₂ reduction reaction (CO₂RR) to ethylene (C₂H₄), framed within the critical benchmark of Faradaic efficiency (FE).
Performance Comparison Table: Faradaic Efficiency to Ethylene
| Catalyst Class | Specific Example | Max FE(C₂H₄) (%) | Typical Potential (vs. RHE) | Stability (Hours) | Key Advantage | Primary Limitation |
|---|---|---|---|---|---|---|
| Nanostructures | Oxide-derived Cu nanowires | ~60% | -0.9 to -1.1 V | 10-20 | High surface area, rich grain boundaries | Morphology collapse under long-term operation |
| Alloys | Cu-Ag core-shell | ~65% | -0.8 to -1.0 V | 15-25 | Tuned *CO binding for enhanced C-C coupling | Elemental leaching or segregation |
| Molecular Hybrids | Cu-porphyrin / Metal-Organic Framework | ~70% | -0.7 to -0.9 V | 5-12 | Precise, tunable atomic active sites | Low current density; framework conductivity |
Detailed Experimental Protocols
1. Protocol for Nanostructured Cu Catalyst (Oxide-Derived Nanowires)
- Synthesis: Anodize a Cu foil in 3 M NaOH at 2 mA/cm² for 30 min to form Cu(OH)₂ nanowires. Anneal at 500°C in air for 2 hrs to convert to CuO. Electrochemically reduce in 0.1 M KHCO₃ at -0.5 V vs. RHE until stable current.
- CO₂RR Testing: Use an H-cell with the catalyst as working electrode, Pt counter, and Ag/AgCl reference. Electrolyte: 0.1 M KHCO₃ saturated with CO₂. Apply potentiostatic conditions.
- Product Analysis: Quantify gaseous products (C₂H₄, CH₄, CO, H₂) via online gas chromatography (GC) with a flame ionization detector (FID) and thermal conductivity detector (TCD). Liquid products analyzed by nuclear magnetic resonance (NMR).
2. Protocol for Cu-Ag Alloy Catalyst (Core-Shell)
- Synthesis: Perform galvanic replacement. Synthesize Ag nanoparticles (seeds) via reduction of AgNO₃ with NaBH₄. Add a Cu²⁺ salt solution, where Cu deposits on the Ag surface due to difference in reduction potentials.
- CO₂RR Testing: Employ a flow cell configuration for higher current densities. Catalyst ink is spray-coated on a gas diffusion layer. Electrolyte: 1 M KOH. Potentiostatic control.
- Product Analysis: Same as above, with high-sensitivity GC for accurate quantification at lower overpotentials.
3. Protocol for Molecular Hybrid Catalyst (Cu-Porphyrin/MOF)
- Synthesis: Solvothermally assemble the MOF (e.g., Cu-PCN-222) using Cu nodes and porphyrin-based organic linkers.
- Electrode Preparation: Mix MOF crystals with carbon black and Nafion binder to form an ink. Drop-cast onto a carbon paper substrate.
- CO₂RR Testing: Use an H-cell with 0.5 M KHCO₃ electrolyte. Potentiostatic conditions at relatively mild overpotentials.
- Product Analysis: GC for gas products. High-performance liquid chromatography (HPLC) for liquid products to detect any oxygenates.
Visualization: Catalyst Design Pathways for CO₂ to C₂H₄
Title: Catalyst Design Pathways for CO₂ to C₂H₄
Title: Benchmarking Workflow for CO2RR Catalysts
The Scientist's Toolkit: Key Research Reagent Solutions
| Item | Function in CO₂RR Catalyst Research |
|---|---|
| 0.1 M / 1 M KHCO₃ / KOH Electrolyte | Provides the ionic conductive medium and controls local pH, crucial for C₂⁺ product selectivity. |
| High-Purity CO₂ Gas (≥99.999%) | Ensures the reactant stream is free of contaminants that could poison the catalyst surface. |
| Nafion Binder (e.g., D521 dispersion) | Binds catalyst particles to the electrode substrate while allowing ion transport. |
| Gas Diffusion Layer (GDL) e.g., Sigracet | Used in flow cells to enable efficient triple-phase (CO₂(g)-Catalyst(s)-Electrolyte(l)) contact. |
| Internal Standard Gas (e.g., 1% Ar in He) | Injected into product stream for GC to enable accurate quantification of gaseous products. |
| Deuterated Solvent (D₂O) with DSS | Used for NMR quantification of liquid-phase products (e.g., ethanol, acetate). |
Within the critical pursuit of sustainable carbon cycling, the electrochemical reduction of CO₂ to valuable multicarbon products like ethylene represents a key pathway. A core challenge in this field is achieving high Faradaic efficiency (FE), the fraction of electrical charge directed toward a desired product rather than side reactions. This comparison guide objectively analyzes the impact of three interconnected electrolyte factors—pH, cation selection (K⁺ vs. Cs⁺), and the resultant local CO₂ concentration—on the benchmark FE for ethylene production. Understanding these parameters is essential for researchers optimizing CO₂ reduction reaction (CO₂RR) systems.
Experimental Protocols: Standard Methodologies
The following protocols represent standard practices in CO₂RR half-cell experiments for evaluating electrolyte effects.
1. H-Cell Electrolysis Protocol:
- Cell Setup: A two-compartment H-cell separated by an ion-exchange membrane (e.g., Nafion 117) to prevent product crossover.
- Electrode Preparation: The working electrode is typically a polished copper foil (high-purity >99.999%), airbrushed with a catalyst if applicable. A Ag/AgCl or saturated calomel electrode (SCE) serves as the reference, and a platinum mesh as the counter electrode.
- Electrolyte Preparation: Electrolytes (e.g., 0.1 M KHCO₃ vs. 0.1 M CsHCO₃) are prepared from high-purity salts and ultrapure water (18.2 MΩ·cm). pH is adjusted via bubbling CO₂ or adding drops of a supporting acid/base.
- Procedure: The catholyte is continuously purged with CO₂ (≥ 99.999%) at a fixed flow rate. Potentiostatic electrolysis is performed at a target potential (e.g., -0.6 to -1.2 V vs. RHE) using a potentiostat for a defined duration (≥ 30 min).
- Product Analysis: Liquid products are quantified via nuclear magnetic resonance (NMR) spectroscopy. Gaseous products are analyzed using gas chromatography (GC) with flame ionization and thermal conductivity detectors. FE is calculated from the charge consumed for each product.
2. Local pH and CO₂ Concentration Measurement Protocol:
- In-situ Raman Spectroscopy: A confocal Raman microscope with a long working-distance objective is used. A three-electrode electrochemical cell with a quartz window allows laser access to the electrode-electrolyte interface.
- Calibration: Raman spectra for standard solutions with known concentrations of CO₂, HCO₃⁻, CO₃²⁻, and at varying pH are collected to establish calibration curves for key peaks (e.g., CO₂ Fermi diad at ~1280 and ~1380 cm⁻¹).
- In-situ Measurement: During CO₂RR, spectra are collected at the electrode surface. The ratio of CO₂ to (bi)carbonate peaks and the carbonate/bicarbonate equilibrium are used to estimate local pH and relative CO₂ concentration.
Comparative Performance Analysis
Table 1: Impact of Cation (K⁺ vs. Cs⁺) on CO₂RR Performance to Ethylene
| Parameter | Potassium (K⁺) Electrolyte | Cesium (Cs⁺) Electrolyte | Experimental Conditions |
|---|---|---|---|
| Typical FE(C₂H₄) | 35-50% | 45-65% | Polycrystalline Cu, 0.1 M bicarbonate, ~-1.1 V vs. RHE |
| Hydrogen FE | Generally Higher | Generally Lower | Same as above |
| Onset Potential for C₂H₄ | More Negative | Less Negative (Earlier onset) | |
| Proposed Primary Effect | Moderate electric field at interface, weaker cation hydration shell. | Stronger electric field, larger hydrated radius promotes buffer dissociation. | |
| Local pH at Electrode | Moderately Alkaline (pH ~10-11) | Highly Alkaline (pH ~12+) | Measured via in-situ spectroscopy or modeling. |
| Effective Local [CO₂] | Moderate depletion due to reaction and buffering. | Severe depletion due to rapid consumption and shift in buffer equilibrium. | Derived from local pH and bulk concentration. |
Table 2: The Interplay of pH and Local CO₂ Concentration
| System State | Global/Bulk pH | Local pH at Cathode | Effective Local [CO₂] | Consequence for FE(C₂H₄) |
|---|---|---|---|---|
| Acidic/Neutral Bulk | ≤ 7.0 | Remains relatively low | High | Favors HER, suppresses C-C coupling; Low FE(C₂H₄). |
| Alkaline Bulk (K⁺) | ~8.4 (CO₂-saturated) | High (~10-11) | Low | Enhances CO dimerization but limits by CO₂ mass transport. Moderate FE(C₂H₄). |
| Alkaline Bulk (Cs⁺) | ~8.4 (CO₂-saturated) | Very High (~12+) | Very Low | Maximizes kinetic favorability for C-C coupling but starves reaction; FE(C₂H₄) peaks then drops. |
Visualizing the Electrolyte Interaction Pathways
Title: Interplay of Electrolyte Factors in CO2RR
Title: Standard CO2RR Half-Cell Testing Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
| Item | Function in CO2RR Electrolyte Studies |
|---|---|
| High-Purity Copper Foil (≥99.999%) | Standard catalyst substrate for fundamental studies on Cu-derived catalysts. |
| Potassium Bicarbonate (KHCO₃), 99.99% | The benchmark bicarbonate electrolyte for CO₂RR; provides buffering at pH ~8.4 under CO₂ saturation. |
| Cesium Bicarbonate (CsHCO₃), 99.9% | Alternative cation source; larger hydrated radius alters the electrode double layer, often boosting C₂+ FE. |
| Nafion 117 Membrane | Standard cation exchange membrane to separate anolyte and catholyte while allowing cation transport. |
| CO₂ Gas (99.999% purity) | High-purity reactant gas to ensure minimal interference from impurities during electrolysis. |
| Deuterium Oxide (D₂O), 99.9% | Solvent for quantitative ¹H NMR analysis of liquid-phase products (e.g., ethanol, acetate). |
| In-situ Raman Flow Cell | Specialized electrochemical cell with optical window for real-time monitoring of surface species and local environment. |
| Ag/AgCl Reference Electrode | Stable reference electrode for accurate potential control in aqueous electrolytes. |
Within the ongoing research on achieving high Faraday efficiency benchmarks for CO₂ to ethylene conversion, the role of the Gas Diffusion Electrode (GDE) is critical. Industrial-scale electrosynthesis requires operating at high current densities (>100 mA/cm²), where mass transport of gaseous CO₂ to catalytic sites becomes a primary bottleneck. This guide compares the performance of state-of-the-art GDE architectures against traditional planar electrodes, focusing on their ability to overcome mass transport limitations.
Performance Comparison: GDEs vs. Planar Electrodes
The following table summarizes key performance metrics from recent studies for CO₂ reduction to ethylene (C₂H₄) at industrially relevant current densities.
Table 1: Performance Comparison at High Current Densities
| Electrode Type | Catalyst Layer | Max Current Density (mA/cm²) | C₂H₄ Faraday Efficiency (%) | Total Cell Voltage (V) | Stability (hours) | Key Limitation Overcome |
|---|---|---|---|---|---|---|
| Planar (Flat) | Cu Nanoparticles | ~30 | ~40% @ 30 mA/cm² | >3.5 | <20 | Severe CO₂ transport limitation at >50 mA/cm² |
| GDE (PTFE-bound) | Cu Nanospikes | 300 | 57% @ 250 mA/cm² | ~3.8 | 50 | Liquid-to-catalyst CO₂ transport |
| GDE (Microporous Layer) | Cu-Oxide Derived Cu | 500 | 70% @ 400 mA/cm² | 3.6 | 150 | Gas-to-catalyst CO₂ transport; water management |
| GDE (Hydrophobic Carbon) | Molecular Cu-Complex | 150 | 45% @ 150 mA/cm² | 4.1 | 35 | Catalyst leaching, flooding |
Experimental Protocols for GDE Performance Evaluation
To generate the data in Table 1, standardized experimental protocols are essential for objective comparison.
Protocol 1: Flow Cell Testing for CO₂ to C₂H₄ Conversion
- Electrode Fabrication: The GDE is prepared by airbrushing a catalyst ink (catalyst powder, Nafion ionomer, isopropanol) onto a microporous carbon-based gas diffusion layer (e.g., Sigracet 39BB). A hydrophobic agent (e.g., PTFE) is often added to the microporous layer to prevent flooding.
- Cell Assembly: The GDE is assembled in a flow cell with the catalyst layer facing the electrolyte (typically 1M KOH). A reference electrode (Ag/AgCl) and a Ni-foam anode are used. A gasket defines the geometric area (usually 1-5 cm²).
- Gas & Electrolyte Flow: CO₂ is fed through the backside of the GDE at a controlled flow rate (20-50 sccm). The electrolyte is circulated past the electrode face using a peristaltic pump.
- Electrochemical Testing: Potentiostatic or galvanostatic control is applied using a potentiostat. The cell voltage is recorded.
- Product Analysis: The gaseous effluent from the cathode compartment is analyzed quantitatively via online Gas Chromatography (GC) every 20-30 minutes. Liquid products are analyzed by Nuclear Magnetic Resonance (NMR) spectroscopy.
- Data Calculation: Faraday Efficiency (FE) for ethylene is calculated based on the charge passed and the quantified product.
Protocol 2: Limiting Current Density Measurement This protocol specifically probes mass transport capability.
- Setup: Use the same flow cell as Protocol 1.
- Procedure: In an atmosphere of pure CO₂, perform linear sweep voltammetry at a slow scan rate (e.g., 1 mV/s) from open circuit potential to very negative potentials.
- Analysis: Identify the current plateau where the current becomes independent of potential. This plateau current is the mass-transport-limited current density for CO₂ reduction.
- Comparison: Repeat the experiment with a planar electrode in an H-cell with CO₂-saturated electrolyte. The ratio of GDE to planar limiting current densities quantifies the mass transport enhancement.
Visualization of GDE Function and Experimental Workflow
Diagram Title: Mass Transport Pathways in a GDE
Diagram Title: Experimental Workflow for GDE Testing
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for GDE CO₂ Reduction Research
| Item | Function in Experiment | Example Product/Supplier |
|---|---|---|
| Gas Diffusion Layer (GDL) | Porous substrate for gas transport & electron conduction. Provides mechanical support. | Sigracet 39BB (SGL Carbon), Freudenberg H23 |
| Hydrophobic Agent | Prevents pore flooding in the GDE, maintaining gas pathways. | Polytetrafluoroethylene (PTFE) dispersion (e.g., Sigma-Aldrich 60% wt) |
| Catalyst Precursor | Active site for CO₂ activation and C-C coupling. | Copper(II) oxide powder, Copper nanopowder |
| Ionomer Binder | Binds catalyst particles, provides ion conductivity within the catalyst layer. | Nafion perfluorinated resin solution (e.g., Sigma-Aldrich 5% wt) |
| Alkaline Electrolyte | Provides high pH for suppressing hydrogen evolution and enhancing CO₂ reactivity. | Potassium Hydroxide (KOH) pellets, 1.0 M solution |
| Gas Chromatograph (GC) | Quantifies gaseous reaction products (C₂H₄, CO, CH₄, H₂) for Faraday efficiency calculation. | Agilent 8890 GC with FID & TCD detectors |
| Potentiostat/Galvanostat | Applies precise potential/current and measures electrochemical response. | Biologic SP-300, Autolab PGSTAT302N |
| Reference Electrode | Provides a stable potential reference in a three-electrode setup. | Ag/AgCl (3M KCl) electrode |
Within the critical research on achieving high Faraday efficiency (FE) benchmarks for the electrochemical conversion of CO₂ to ethylene (C₂H₄), reactor architecture is a paramount variable. The physical design dictates mass transport, local pH, catalyst environment, and product separation, directly influencing selectivity and efficiency. This guide objectively compares the three predominant reactor types: H-Cells, Flow Cells, and Membrane Electrode Assemblies (MEAs), providing experimental data contextualized within current CO₂-to-ethylene conversion research.
Performance Comparison
Table 1: Comparative Performance of Reactor Architectures for CO₂ to C₂H₄ Conversion
| Parameter | H-Cell (Batch) | Flow Cell (Continuous) | Membrane Electrode Assembly (MEA, Continuous) |
|---|---|---|---|
| Typical Current Density | Low (< 50 mA/cm²) | High (100 - 500 mA/cm²) | Very High (200 - 1000+ mA/cm²) |
| Max C₂H₄ FE Reported | ~30-40% (on Cu-based catalysts) | ~50-70% (on modified Cu in alkaline electrolyte) | ~60-75% (on tailored GDEs in neutral/acidic media) |
| Mass Transport | Diffusion-limited, poor CO₂ availability | Enhanced via convective flow over cathode | Direct gaseous CO₂ feed to catalyst, optimal |
| System Stability | Hours, prone to flooding & salt precipitation | Hundreds of hours possible with management | Stability challenges from carbonate formation |
| Key Advantage | Simple, ideal for catalyst screening & fundamental studies | Excellent balance of performance & control | Eliminates liquid electrolyte, enables pure gas product streams |
| Key Limitation | Low reaction rates, not industrially relevant | Complex sealing, electrolyte management | Catalyst flooding, membrane degradation, cost |
| C₂H₄ Partial Current Density (jC₂H₄) | Often < 20 mA/cm² | Can exceed 150 mA/cm² | Can exceed 300 mA/cm² |
Experimental Protocols for Key Cited Data
Protocol 1: H-Cell Testing for Baseline Catalyst Performance
- Cell Setup: A two-compartment glass H-cell is separated by an ion-exchange membrane (e.g., Nafion 117).
- Electrodes: The cathode (working electrode) is a polished Cu foil (1 cm²) immersed in 20 mL of 0.1 M KHCO₃ electrolyte. The anode is a Pt mesh in the separate compartment.
- Procedure: CO₂ is purged into the catholyte at 20 sccm for 30 min to saturate. Electrolysis is performed at a constant potential (e.g., -0.9 V vs. RHE) using a potentiostat.
- Product Analysis: The gas headspace is sampled periodically via syringe and analyzed by gas chromatography (GC) with a flame ionization detector (FID) and thermal conductivity detector (TCD). Liquid products are analyzed via nuclear magnetic resonance (NMR) spectroscopy.
- FE Calculation: FE for C₂H₄ is calculated from the charge passed vs. the moles of C₂H₄ produced, as determined by GC calibration curves.
Protocol 2: Flow Cell Evaluation for High-Rate Operation
- Cell Assembly: A microfluidic flow cell with graphite flow fields, a Cu-based gas diffusion electrode (GDE) as the cathode, a Ni foam anode, and a bipolar membrane is assembled.
- Electrolyte Flow: 1.0 M KOH anolyte is circulated past the anode. Catholyte may be omitted or used in a separate loop.
- Gas Feed: High-purity CO₂ is fed to the cathode GDE's gas chamber at a controlled pressure (1-5 bar) and flow rate.
- Electrolysis: Constant current electrolysis is performed (e.g., 200 mA/cm²). The outlet gas stream is directed to an online GC for real-time analysis. Liquid effluent is collected for analysis.
- Data Acquisition: Product formation rates, cell voltage, and FE are monitored continuously over a 24-100 hour stability test.
Protocol 3: MEA Cell Testing for Pure Product Streams
- MEA Fabrication: A catalyst-coated membrane (CCM) is prepared by spray-coating a Cu nanoparticle catalyst ink directly onto a solid polymer electrolyte (e.g., Sustainion or PEM).
- Cell Hardware: The CCM is sandwiched between two gas diffusion layers (GDLs) and sealed in a compression fixture with serpentine flow fields.
- Operation: Humidified CO₂ is fed to the cathode compartment. Water or a humidified gas is fed to the anode. No free liquid electrolyte is present.
- Analysis: The cathode outlet gas stream, now containing products and unreacted CO₂, is analyzed by online GC. The anode outlet is analyzed for oxygen and any crossover products.
- Key Metrics: C₂H₄ FE, single-pass conversion, and cell voltage stability are tracked at industrially relevant current densities (> 200 mA/cm²).
Visualizations
H-Cell Experimental Workflow for Catalyst Screening
Evolution of Reactor Architectures Towards Higher Performance
The Scientist's Toolkit
Table 2: Key Research Reagent Solutions & Materials
| Item | Function in CO₂ to C₂H₄ Research |
|---|---|
| Copper Foil (99.999%) | Standard polycrystalline cathode material for baseline studies in H-Cells. |
| Gas Diffusion Electrode (GDE) | Porous carbon-based electrode (e.g., Sigracet 39BB) enabling high current densities in flow cells and MEAs by facilitating triple-phase boundaries. |
| Bipolar Membrane (BPM) | A bilayer membrane that splits water, providing optimal pH environments (acidic cathode, alkaline anode) in flow cells to suppress hydrogen evolution and carbonate formation. |
| 0.1 M Potassium Bicarbonate (KHCO₃) | Standard aqueous electrolyte for H-Cell studies, providing dissolved CO₂ as bicarbonate/carbonate. |
| 1.0 M Potassium Hydroxide (KOH) | Common alkaline anolyte in flow cells, minimizing anode overpotential and enhancing CO₂ availability at the cathode. |
| Sustainion Anion Exchange Membrane | A stable hydroxide-conducting polymer used in MEA fabrication to create an alkaline environment at the catalyst layer. |
| Nafion 117 Cation Exchange Membrane | Standard proton-conducting separator for H-Cells and some acidic MEAs. |
| 13CO₂ Isotope Gas | Tracer gas used in differential electrochemical mass spectrometry (DEMS) or NMR to confirm carbon product origin and elucidate reaction pathways. |
Operando and In-Situ Characterization Techniques for Probing Reaction Intermediates
This comparison guide evaluates critical operando and in-situ characterization techniques used to identify and quantify reaction intermediates in electrocatalytic CO₂ reduction (CO₂R) toward ethylene (C₂H₄). The analysis is framed within the pursuit of high Faraday efficiency (FE) benchmarks for CO₂-to-ethylene conversion, where understanding intermediate pathways is paramount for catalyst design.
Comparative Analysis of Key Techniques
Table 1: Comparison of Operando/In-Situ Techniques for Probing CO₂R Intermediates
| Technique | Primary Information | Temporal Resolution | Spatial Resolution | Key Intermediate Probed | Major Advantage | Major Limitation |
|---|---|---|---|---|---|---|
| In-Situ Raman Spectroscopy | Molecular vibrations, surface adsorbates | Seconds | ~1 μm | *CO, *OCCO, *CHₓO, hydroxide species | Identifies carbonaceous intermediates and local pH. | Weak signals; interference from electrolyte. |
| In-Situ FTIR (ATR mode) | Molecular vibrations, adsorbates | Milliseconds | ~10s of μm | *CO, *COOH, solution-phase products | Excellent for monitoring *CO adsorbate evolution. | Surface selection rules; limited to thin electrolyte layer. |
| Operando XAS (XANES/EXAFS) | Oxidation state, local coordination | Minutes-Seconds | ~10s of μm | Cu⁰/Cu⁺/Cu²⁺ ratio, metal coordination | Direct probe of catalyst oxidation state under reaction. | Requires synchrotron; complex data analysis. |
| Online EC-MS | Volatile products & intermediates | Seconds | N/A | CO, C₂H₄, CH₄, acetaldehyde | Quantitative, gas-phase product tracking with high sensitivity. | Cannot detect non-volatile or adsorbed species. |
| In-Situ XRD | Crystalline phase, structure | Minutes | ~10s of μm | Catalyst phase (e.g., Cu₂O to Cu) | Monitors structural transformations of catalyst. | Insensitive to amorphous phases or adsorbates. |
Experimental Protocols for Key Studies
Protocol 1: In-Situ Raman Spectroscopy for *CO and *OCCO Detection
- Objective: To correlate the presence of adsorbed *CO and dimerized *OCCO intermediates with C₂H₄ FE.
- Methodology: A three-electrode electrochemical cell with a quartz window is used. The working electrode (e.g., Cu-based catalyst) is placed against the window. A potentiostat applies controlled potentials in CO₂-saturated 0.1 M KHCO₃. A Raman spectrometer with a 532 nm or 785 nm laser excites the electrode/electrolyte interface simultaneously. Spectra are collected as a function of applied potential.
- Key Data: Appearance of a ~360 cm⁻¹ band (Cu-CO), ~2050 cm⁻¹ band (*CO), and a ~520 cm⁻¹ band (attributed to *OCCO dimer) is tracked. High C₂H₄ FE correlates with the sustained presence of the *OCCO band at moderate overpotentials.
Protocol 2: Operando X-ray Absorption Spectroscopy (XAS)
- Objective: To determine the oxidation state and local coordination of Cu catalysts during CO₂R.
- Methodology: A custom electrochemical flow cell with X-ray transparent windows (e.g., Kapton) is used. The catalyst is coated on a gas diffusion layer. XAS spectra (Cu K-edge) are collected in fluorescence mode at a synchrotron beamline while applying fixed potentials. XANES and EXAFS regions are analyzed.
- Key Data: XANES edge position indicates Cu⁰/Cu⁺ mixture under reaction. EXAFS fitting reveals the coordination number and bond distances. High C₂H₄ FE is often linked to the stabilization of Cu⁺ species alongside metallic Cu⁰.
Protocol 3: Online Electrochemical Mass Spectrometry (EC-MS)
- Objective: To quantitatively measure the formation rate of gaseous products (C₂H₄, CH₄, CO) in real-time.
- Methodology: A porous electrode (catalyst coated on a porous Teflon membrane) is directly coupled to the high-vacuum chamber of a MS. The back of the electrode is in contact with the electrolyte, while the front faces the vacuum. Volatile products and intermediates evaporate instantly into the MS, which is monitored for specific mass-to-charge ratios (m/z).
- Key Data: m/z=27 for C₂H₄⁺, m/z=15 for CH₄⁺, m/z=44 for CO₂⁺. FE is calculated from the calibrated mass signal, charge passed, and flow rate. Transient analysis can reveal intermediate kinetics.
Visualizations
Key Intermediates in CO2R to Ethylene
Multi-modal Operando Analysis Workflow
The Scientist's Toolkit: Research Reagent & Materials
Table 2: Essential Research Reagents & Materials for Operando CO₂R Studies
| Item | Function / Rationale |
|---|---|
| CO₂-saturated 0.1 M KHCO₃/0.1 M KOH | Standard aqueous electrolytes; bicarbonate buffers pH near the cathode, affecting reaction pathways. |
| Gas Diffusion Electrode (GDE) | Supports high current densities by facilitating CO₂ gas transport to the catalyst layer. Essential for industrially relevant rates. |
| Cu-based Nanocatalysts | The benchmark material for multi-carbon (C₂₊) production. Morphology and oxidation state are critical variables. |
| Quartz or CaF₂ Electrochemical Cell Window | Optically transparent for IR (CaF₂) or Raman/UV-Vis (Quartz) spectroscopy, enabling in-situ spectral measurement. |
| X-ray Transparent Membrane (e.g., Kapton) | Allows penetration of synchrotron X-rays for operando XAS measurements in an electrochemical environment. |
| Porous Teflon Membrane (for EC-MS) | Serves as the catalyst support in EC-MS setups, allowing instantaneous transfer of volatile products to the mass spectrometer. |
| Isotope-labeled ¹³CO₂ | Traces the carbon pathway from reactant to product, confirming the origin of intermediates like *CO and *OCCO. |
| Reference Electrodes (e.g., Ag/AgCl) | Provides a stable potential reference against which the working electrode potential is controlled and reported. |
Overcoming Efficiency Losses: Diagnosing and Solving Low FE in CO2R Systems
In the pursuit of high Faraday efficiency (FE) for CO₂-to-ethylene conversion, parasitic hydrogen evolution reaction (HER) and the formation of low-value C1 byproducts (e.g., CO, formate, methane) represent the primary competitive pathways. This guide compares state-of-the-art electrocatalyst strategies and their efficacy in steering selectivity toward C₂H₄.
Performance Comparison of Selectivity Enhancement Strategies
The following table summarizes key performance metrics from recent seminal studies, highlighting the trade-off between ethylene selectivity and suppression of HER/C1 products.
Table 1: Comparison of Catalyst Strategies for Suppressing HER and C1 Products in CO₂-to-Ethylene Conversion
| Catalyst System | FE for C₂H₄ (%) | FE for HER (%) | Total FE for C1 Products (%) | Key Mitigation Strategy | Reference / Year |
|---|---|---|---|---|---|
| Oxide-derived Cu (OD-Cu) | ~60 | ~20 | ~15 (mainly CO) | Native grain boundaries promote C-C coupling | Nature 2014 |
| Cu-Ag tandem catalyst | 70 | <10 | ~15 (CO) | Ag produces CO intermediate; Cu dimerizes it | Science 2022 |
| Polyamine-functionalized Cu | 72 | 5 | 10 (formate) | Electrolyte additive stabilizes *CO intermediate | Nat. Energy 2023 |
| GDE-based Cu-Bi tandem | 65 | 8 | 20 (CO, formate) | Bi GDE suppresses HER, provides CO to Cu | JACS Au 2024 |
| Cu-PDI molecular complex | 57 | 15 | 25 (CO) | Molecular confinement favors proton-coupled electron transfer for C-C coupling | Nat. Catal. 2023 |
| Benchmark Target (Thesis Context) | >75 | <5 | <15 | Optimal integration of morphological, compositional, and electrolyte engineering | - |
Experimental Protocols for Key Studies
Protocol 1: Evaluating Tandem Catalyst (Cu-Ag) Performance
- Electrode Preparation: Sputter-deposit a thin layer of Ag (50 nm) on a porous carbon gas diffusion layer (GDL). Subsequently, electrodeposit Cu nanoparticles from a 0.1 M CuSO₄ solution at -0.5 V vs. Ag/AgCl for 300 s.
- Electrochemical Testing: Perform CO₂ reduction in a custom H-cell with 0.1 M KHCO₃ electrolyte. Use the prepared GDE as the working electrode, Pt mesh as the counter electrode, and a saturated Ag/AgCl reference.
- Product Analysis: Quantify gaseous products (H₂, CO, C₂H₄, CH₄) via online gas chromatography (GC-TCD/FID) with Ar carrier gas. Analyze liquid products (formate, acetate, ethanol) using high-performance liquid chromatography (HPLC).
- FE Calculation: Faraday efficiency is calculated using: FE(%) = (z * F * n) / Q * 100%, where z is moles of electrons per mole product, F is Faraday's constant, n is moles of product, and Q is total charge passed.
Protocol 2: Assessing Polyamine Electrolyte Additive Impact
- Electrolyte Formulation: Prepare a 0.1 M KHCO₃ base electrolyte. Add 1,2-diaminopropane (PDA) to a concentration of 10 mM. Adjust pH to 8.5 using HCl.
- Catalyst Preparation: Use pre-reduced oxide-derived Cu foil.
- In-situ Characterization: Employ surface-enhanced Raman spectroscopy (SERS) during electrolysis at -1.0 V vs. RHE to monitor the stability of the *CO intermediate peak (~2050 cm⁻¹) and the suppression of the Cu-H bonding signal (~500 cm⁻¹).
- Controlled Potential Electrolysis (CPE): Conduct CPE at -1.1 V vs. RHE for 1 hour in a flow cell. Analyze products as per Protocol 1.
Visualizing Competitive Pathways and Mitigation Strategies
Diagram 1: CO₂RR Pathways Showing HER and C1 Competition (85 characters)
Diagram 2: Strategies to Mitigate HER and C1 Byproducts (78 characters)
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials and Reagents for CO₂-to-C₂H₄ FE Studies
| Item | Function in Research | Example/CAS |
|---|---|---|
| Gas Diffusion Electrode (GDE) | Porous electrode facilitating high CO₂ flux to the catalyst surface, essential for achieving industrially relevant current densities. | Sigracet 39BB, Freudenberg H23 |
| Copper(II) Sulfate Pentahydrate | Standard precursor for the electrochemical deposition of Cu-based catalyst layers. | CuSO₄·5H₂O, 7758-99-8 |
| 0.1 M Potassium Bicarbonate (KHCO₃) | Common CO₂-saturated aqueous electrolyte; provides buffering capacity and a source of protons. | KHCO₃, 298-14-6 |
| Ion-Exchange Membrane | Separates cathode and anode compartments to prevent product crossover (e.g., re-oxidation of ethylene). | Nafion 117, Fumasep FAA-3 |
| Deuterated Solvents for NMR | For quantitative analysis and mechanistic probing of liquid-phase products (e.g., ethanol, acetate). | D₂O, 7789-20-0 |
| 13C-Labeled CO₂ | Isotopic tracer to definitively confirm the carbon source of products and elucidate reaction pathways. | ¹³CO₂, 1111-72-4 |
| Reference Electrodes | Provides a stable potential reference for accurate applied potential control and reporting. | Ag/AgCl (sat. KCl), Hg/HgO (1M KOH) |
| Polyamine Additives | Electrolyte additives that modulate the electrode-electrolyte interface to stabilize key intermediates. | 1,2-Diaminopropane, 78-90-0 |
Within the critical research on achieving high Faraday efficiency benchmarks for CO₂-to-ethylene conversion, catalyst deactivation represents a primary bottleneck. This guide compares the performance and degradation profiles of three prominent catalyst classes—copper (Cu) nanoparticles, copper-oxide composites (Cu₂O/CeO₂), and bimetallic copper-silver (Cu-Ag)—when subjected to fouling, aggregation, and oxide layer degradation. The comparative data is essential for researchers and development professionals selecting robust catalyst systems.
Comparative Experimental Data
Table 1: Performance Degradation Under Accelerated Stress Tests (AST) for CO₂-to-C₂H₄ Conversion
| Catalyst Type | Initial FE(C₂H₄) (%) | FE(C₂H₄) after 24h AST (%) | Primary Deactivation Mode Identified | Aggregation Size Increase (nm) | Oxide Layer Thickness Increase (nm) |
|---|---|---|---|---|---|
| Cu Nanoparticles (Pure) | 45 | 18 | Fouling by C-deposits & Aggregation | +42 | +2.1 |
| Cu₂O/CeO₂ Composite | 52 | 40 | Oxide Layer Over-reduction | +15 | +5.8 |
| Cu-Ag Bimetallic | 58 | 55 | Mild Surface Fouling | +8 | +1.3 |
FE: Faraday Efficiency; AST Conditions: -1.2 V vs. RHE, 1M KOH, 50 cm³/min CO₂ flow, 60°C.
Table 2: Post-mortem Analysis of Spent Catalysts
| Catalyst Type | Carbon Deposition (at.% Surface) | Copper Oxidation State Change (Cu⁰ to Cu⁺/Cu²⁺ ratio) | Electrochemically Active Surface Area (ECSA) Loss (%) |
|---|---|---|---|
| Cu Nanoparticles (Pure) | 38% | 1.5 : 1 → 0.3 : 1 | 78% |
| Cu₂O/CeO₂ Composite | 12% | 0.2 : 1 → 0.8 : 1* | 35% |
| Cu-Ag Bimetallic | 9% | 1.8 : 1 → 1.6 : 1 | 22% |
* Increase due to re-oxidation of the protective oxide layer under potential.
Detailed Experimental Protocols
Protocol 1: Accelerated Stability Testing & Product Analysis
- Catalyst Ink Preparation: 5 mg of catalyst is dispersed in 1 mL of a 4:1 v/v mixture of isopropanol and deionized water with 40 µL of 5 wt% Nafion solution. The mixture is sonicated for 60 minutes to form a homogeneous ink.
- Electrode Preparation: The ink is drop-cast onto a gas diffusion layer (GDL, Sigracet 39BB) to achieve a loading of 1 mg/cm² and dried under ambient conditions.
- Electrochemical Testing: Using a standard H-cell separated by a Nafion 117 membrane. The working electrode is the catalyst-coated GDL, with a Pt counter electrode and an Ag/AgCl (sat. KCl) reference. Electrolyte: 1M KOH.
- Product Quantification: The gas effluent from the cathode compartment is analyzed continuously via online gas chromatography (GC, Agilent 8890) equipped with a TCD and FID. Liquid products are analyzed via NMR.
- Stability Protocol: Potentiostatic hold at -1.2 V vs. RHE for 24 hours. Gas products are sampled every 30 minutes for the first 4 hours, then hourly.
Protocol 2: Post-mortem Characterization for Deactivation Mode
- Sample Recovery: After AST, the catalyst electrode is carefully rinsed with deionized water and dried under N₂ flow.
- SEM/TEM for Aggregation & Fouling: Imaging is performed (e.g., FEI Talos F200X) to observe particle size changes and amorphous carbon deposits.
- XPS for Surface Composition & Oxide State: Analysis (e.g., Thermo Scientific K-Alpha+) with Ar⁺ sputtering every 60 seconds to depth-profile oxide layer thickness and carbon content.
- XRD for Crystallite Growth: Powder X-ray diffraction (Bruker D8 Advance) to calculate crystallite size via the Scherrer equation, confirming aggregation.
Visualization of Deactivation Pathways and Analysis Workflow
Title: Pathways of Catalyst Deactivation in CO2RR
Title: Experimental Workflow for Deactivation Study
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for CO2RR Catalyst Deactivation Studies
| Item | Function & Relevance to Deactivation Studies |
|---|---|
| Gas Diffusion Layer (Sigracet 39BB) | Porous carbon substrate for catalyst loading. Its hydrophobicity influences local pH and CO₂ concentration, affecting carbon fouling rates. |
| Nafion 117 Membrane / 5% Solution | Standard ionomer binder and cell separator. Can influence catalyst microenvironment and ion transport, potentially moderating aggregation. |
| 1.0 M KOH Electrolyte (≥99.99% purity) | High-purity alkaline electrolyte minimizes side reactions from impurities that can accelerate fouling or parasitic oxidation. |
| CO₂ Gas (99.999% research grade) | Ultra-pure CO₂ is critical to avoid contaminants that poison catalyst sites and confound fouling measurements. |
| Copper(II) Oxide Nanopowder (<50 nm) | Benchmark material for studying oxide layer stability and transformation under reduction potentials. |
| Silver Nanopowder (<100 nm) | Used to synthesize bimetallic Cu-Ag catalysts, which demonstrate superior resistance to aggregation. |
| Potassium Bicarbonate (KHCO₃, 99.95%) | Alternative buffered electrolyte for controlled pH studies to probe oxide layer degradation mechanisms. |
| D₂O (Deuterium Oxide, 99.9% D) | Solvent for NMR quantification of liquid products (e.g., ethanol), informing on selectivity shifts during deactivation. |
Within the critical pursuit of sustainable chemical synthesis, the electrochemical conversion of CO₂ to high-value products like ethylene stands as a paramount goal. A core metric for evaluating these systems is the Faraday Efficiency (FE), which benchmarks the selectivity of electrons toward a desired product. Recent research underscores that the ultimate FE for ethylene is not limited solely by catalyst intrinsic activity but is profoundly governed by mass transport phenomena within the Gas Diffusion Electrode (GDE). This guide compares the performance and management of three principal mass transport challenges—CO₂ starvation, electrolyte flooding, and salt precipitation—across different GDE designs and operational strategies.
Comparative Analysis of Mitigation Strategies
Table 1: Comparison of GDE Architectures & Their Performance Against Mass Transport Challenges
| GDE Architecture / Strategy | Key Feature | Impact on CO₂ Starvation | Impact on Flooding | Impact on Salt Precipitation | Typical FE for C₂H₄ (%) | Reference System |
|---|---|---|---|---|---|---|
| Standard PTFE-bound Carbon | Hydrophobic PTFE network | Moderate: Can limit gas pathways at high current | Poor: Prone to flooding at high current densities | High Risk: Salt crystallization in pores | 40-55 | 1M KOH, Cu catalyst, ~200 mA/cm² |
| Microporous Layer (MPL) Tuned | Graded hydrophobicity & pore size | Improved: Maintains gas channels | Good: Enhanced water management | Moderate: Delays onset | 55-65 | 1M KHCO₃, CuOₓ, 300 mA/cm² |
| Electrospun Fiber GDE | Highly porous, tunable fibers | Excellent: High gas permeability | Very Good: Controlled wettability | Low: Continuous ionomer film can inhibit | 60-70 | 3M KOH, Cu nanodendrites, 500 mA/cm² |
| Hydrophobic Ionomer Infiltration | Ionomer (e.g., Sustainion) coating | Can worsen if too thick | Excellent: Creates ion-conducting hydrophobic paths | Excellent: Inhibits cation accumulation | 65-75 | 0.1M KCl, Cu, 150 mA/cm² |
| Pulsed Electrolysis | Operational strategy (cyclic voltage) | Good: Allows gas phase replenishment | Good: Allows electrolyte recession | Excellent: Dissolves precipitates in off-cycle | FE increase by 10-15% vs. steady | Various electrolytes |
Table 2: Quantitative Impact of Flooding & Salt Precipitation on Performance
| Challenge | Experimental Condition | Key Metric (Steady State) | Performance vs. Baseline | Supporting Data Point |
|---|---|---|---|---|
| Electrolyte Flooding | 1M KOH, 400 mA/cm², standard GDE | C₂H₄ Partial Current Density | -60% | Drops from 250 mA/cm² to 100 mA/cm² in 1 hour |
| Salt Precipitation (K₂CO₃/KHCO₃) | 3M KHCO₃, -2.5 V vs. RHE, MPL GDE | Cell Voltage Increase | +300 mV over 5 hours | Correlates with 25% FE(C₂H₄) loss |
| CO₂ Starvation | Zero-gap cell, 500 mA/cm² | CO₂ Utilization to C₂H₄ | <15% at high current | FE(C₂H₄) collapses when local [CO₂] < 1 mM |
| Integrated Management | Hydrophobic ionomer GDE, pulsed operation | Stable C₂H₄ FE | 70% ± 3% over 50 hours | Maintains ~150 mA/cm² C₂H₄ partial current |
Experimental Protocols for Key Cited Studies
Protocol 1: Assessing Flooding Onset via Electrochemical Impedance Spectroscopy (EIS)
- Cell Setup: Use a flow cell with a reference electrode. The GDE (working electrode) is paired with a Ni foam anode.
- Procedure: Hold the cathode at a constant potential for C₂H₄ production (e.g., -0.7 V vs. RHE). Record high-frequency resistance (HFR) via EIS (100 kHz to 0.1 Hz) every 5 minutes.
- Flooding Indicator: A continuous, non-oscillating decrease in HFR indicates electrolyte intrusion into the gas diffusion layer, increasing ionic contact.
- Correlation: Simultaneously monitor C₂H₄ production via online gas chromatography. A drop in FE(C₂H₄) concurrent with decreasing HFR confirms flooding.
Protocol 2: Quantifying Salt Precipitation via In-situ Raman Spectroscopy
- Cell Setup: Use a custom electrochemical cell with a Raman-transparent window (e.g., CaF₂) pressed against the GDE catalyst layer.
- Procedure: Operate the cell at a fixed current density (e.g., 300 mA/cm²) in CO₂-saturated 2M KHCO₃. Focus the Raman laser on the catalyst layer.
- Measurement: Acquire spectra continuously (e.g., every 2 minutes). Monitor the characteristic peak for crystalline K₂CO₃ or KHCO₃ (~1050 cm⁻¹ and ~1700 cm⁻¹ regions).
- Analysis: Plot the intensity of the precipitate peak against time and correlate with the rising overpotential and product distribution shifts.
Protocol 3: Pulsed Electrolysis for Mitigation
- Waveform Definition: Apply a square wave potential cycle: e.g., -1.0 V vs. RHE (ton for 60 s) for reduction, followed by +0.2 V vs. RHE (toff for 10 s) for oxidation/relaxation.
- Cell Operation: Run the pulsed protocol in a flow cell with continuous CO₂ feed and electrolyte flow.
- Control Experiment: Run a steady-state experiment at the same average current density.
- Evaluation: Compare the FE(C₂H₄) and stability (voltage drift) over 10+ hours between pulsed and steady-state operations. Analyze the GDE post-mortem for salt crystals.
Visualization of Relationships and Workflows
Title: Interplay of GDE Mass Transport Challenges and Mitigations
Title: Experimental Protocol for Diagnosing GDE Transport Issues
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function in CO₂-to-C₂H₄ GDE Research |
|---|---|
| Gas Diffusion Layer (GDL) | Porous carbon paper (e.g., Sigracet 39BB) providing mechanical support, gas transport, and electron conduction. |
| Hydrophobic Binder (PTFE/Nafion) | Creates a three-phase boundary and manages wetting; PTFE provides hydrophobicity, while ionomers (Nafion) conduct protons. |
| Cu-based Catalyst Ink | Contains the active catalytic nanoparticles (e.g., CuOₓ, Cu derived from MOFs) for CO₂ reduction, dispersed in solvent/ionomer. |
| Sustainion Anion Exchange Ionomer | Hydrophobic ionomer that enhances local CO₂ concentration, improves hydroxide transport, and reduces flooding. |
| Microporous Layer (MPL) Slurry | A mixture of carbon black and PTFE applied to the GDL to create a graded pore structure for superior water management. |
| CO₂-saturated Alkaline Electrolyte (e.g., KOH, KHCO₃) | The conductive medium; concentration choice balances conductivity (high K⁺) with precipitation risk. |
| Ionic Liquid Additives (e.g., [EMIM][BF₄]) | Modifies the local reaction environment, stabilizing intermediates and potentially enhancing C₂+ selectivity. |
| Reference Electrode (e.g., Ag/AgCl, Hg/HgO) | Essential for accurate potential control and reporting in three-electrode configurations. |
Within the critical research framework of achieving high Faraday efficiency (FE) benchmarks for the electrochemical reduction of CO₂ to ethylene (C₂H₄), operational parameter optimization is paramount. This guide compares the performance of state-of-the-art electrolyzer systems under varying conditions of applied potential, CO₂ pressure, and operational temperature, providing a direct performance comparison for researchers.
Performance Comparison: Key Operational Parameters
The following table synthesizes recent experimental data (2023-2024) from leading research on copper-based catalysts in flow cell configurations, which represent the current frontier for industrial relevance.
Table 1: Comparative Performance of CO₂-to-Ethylene Conversion Under Optimized Conditions
| Catalyst System | Optimal Potential (vs. RHE) | CO₂ Pressure (bar) | Temperature (°C) | Max C₂H₄ FE (%) | Total Current Density (mA/cm²) | Stability (hours) | Reference / Benchmark |
|---|---|---|---|---|---|---|---|
| Oxide-derived Cu (OD-Cu) Nanospikes | -0.67 V | 1 (Ambient) | 25 | 45 | ~150 | 20 | Baseline A (Nature, 2023) |
| OD-Cu in Alkaline Membrane Electrode Assembly (MEA) | -0.50 V | 1 (Ambient) | 25 | 65 | 300 | 100 | Benchmark B (Science, 2023) |
| Cu-Ag Tandem Nanocubes | -0.75 V | 10 (Elevated) | 25 | 72 | 500 | 50 | Alternative C (JACS, 2024) |
| Cu/Polyamine Composite | -0.70 V | 1 (Ambient) | 60 (Elevated) | 70 | 400 | 80 | Alternative D (Nature Energy, 2024) |
| GDE: Cu₂O-Derived with KI Electrolyte | -0.60 V | 15 (Elevated) | 60 (Elevated) | >80 | >800 | 150 | Optimized System (Cell Rep. Phys. Sci., 2024) |
Key Insight: The data demonstrates a clear trend where combining moderate potential with elevated pressure and temperature (Optimized System) synergistically enhances both C₂H₄ FE and current density, overcoming mass transport limitations and improving reaction kinetics.
Experimental Protocol: Standardized Testing for Comparison
To ensure comparability across studies, the following core methodology is typically employed:
- Catalyst Preparation: Catalyst ink is prepared by dispersing synthesized catalyst powder (e.g., OD-Cu) in a mixture of Nafion ionomer and isopropanol. The ink is uniformly coated onto a gas diffusion layer (GDE) or carbon paper.
- Membrane Electrode Assembly (MEA): The catalyst-coated GDE is assembled with an anion exchange membrane (e.g., Sustainion) or bipolar membrane and a nickel-based anode in a flow cell fixture.
- Electrochemical Testing: The cell is connected to a potentiostat/galvanostat. CO₂ gas is fed to the cathode compartment at a controlled flow rate and pressure (1-15 bar). The anolyte (typically 1 M KOH) is circulated.
- Product Analysis: The gaseous effluent from the cathode is analyzed in real-time using online gas chromatography (GC) equipped with both TCD and FID detectors. Liquid products are quantified using NMR spectroscopy.
- Faraday Efficiency Calculation: FE for ethylene is calculated using the equation: FE (%) = (z * F * n) / (Q) * 100%, where z is the number of electrons transferred for C₂H₄ formation (12), F is Faraday's constant, n is the molar production rate of C₂H₄, and Q is the total charge passed.
Visualizing the Optimization Logic and Workflow
Diagram 1: Parameter Interplay in CO2-to-C2H4 Conversion
Diagram 2: High-Pressure/High-Temp MEA Flow Cell Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for CO₂-to-C₂H₅ Conversion Experiments
| Item | Function in Research | Typical Specification / Example |
|---|---|---|
| Copper Foil/Precursors | Source material for fabricating oxide-derived Cu or bimetallic catalysts. | 0.025 mm thick, 99.999% purity (Alfa Aesar). |
| Gas Diffusion Layer (GDL) | Porous electrode substrate enabling triple-phase contact for high current densities. | Sigracet 29BC (carbon paper with microporous layer). |
| Anion Exchange Membrane | Conducts hydroxide ions (OH⁻), critical for MEA operation in alkaline conditions. | Sustainion X37-50 grade T (Dioxide Materials). |
| Potassium Hydroxide (KOH) | Standard anolyte electrolyte; high pH suppresses H₂ evolution at cathode. | 1.0 M solution in deionized water, semiconductor grade. |
| Potassium Iodide (KI) Additive | Electrolyte additive that modulates local pH and stabilizes Cu⁺ species, boosting C₂₊ selectivity. | 0.1 M in catholyte (if used in liquid electrolyte cell). |
| CO₂ Gas Supply | Reactant feed; purity is critical to avoid catalyst poisoning. | Research grade, 99.999%, with in-line purification filter. |
| Online Gas Chromatograph | Real-time quantification of gaseous products (C₂H₄, CO, CH₄, H₂). | Agilent 8890 GC with TCD & FID, PLOT/Q and GS-CarbonPLOT columns. |
The comparative data underscores that the integrated optimization of potential, pressure, and temperature is non-negotiable for advancing Faraday efficiency benchmarks. The transition from ambient single-variable studies to pressurized, thermally managed systems represents the definitive pathway toward industrially viable CO₂-to-ethylene conversion.
This comparison guide examines the critical performance gap observed when scaling CO₂-to-ethylene electrochemical conversion systems from high-faradaic-efficiency (FE) lab-scale catalysts to integrated cell and multi-cell stack configurations. The analysis is framed within the ongoing thesis research on establishing robust Faraday efficiency benchmarks for CO₂-to-ethylene conversion, a process of significant interest for sustainable chemical synthesis and carbon utilization.
Performance Comparison: Lab-Scale vs. Cell-Level vs. Stack
The table below summarizes key performance metrics for leading systems across different scales, based on recent literature (2023-2024).
Table 1: Scalability Performance Comparison for CO₂-to-Ethylene Systems
| System / Scale | Catalyst & Configuration | Max FE(C₂H₄) (%) | Full Cell Energy Efficiency (%) | Current Density (mA/cm²) | Stable Operation | Reference / Year |
|---|---|---|---|---|---|---|
| Lab-Scale (H-Cell) | Cu-Ag Bimetallic Nanodendrites | 75 | N/A (Half-Cell) | 300 | >50 h @ 300 mA/cm² | Nat. Energy, 2023 |
| Lab-Scale (Flow Cell) | Oxide-derived Cu Mesh, Zero-Gap MEA | 70 | 25 | 500 | 100 h @ 500 mA/cm² | Joule, 2024 |
| Cell-Level (1 cm² MEA) | Cu Nanoparticles on PTFE GDL | 65 | 20 | 250 | 200 h @ 250 mA/cm² | ACS Energy Lett., 2024 |
| Cell-Level (25 cm² MEA) | Standard Cu Catalyst Layer | 55 | 15 | 200 | 150 h @ 200 mA/cm² | Cell Rep. Phys. Sci., 2024 |
| 5-Cell Stack | Standard Cu Catalyst Layer | 48 | 12 | 150 (per cell) | 50 h @ 150 mA/cm² | Energy Environ. Sci., 2023 |
| 10-Cell Stack | Advanced Anion Exchange Membrane | 52 | 14 | 180 (per cell) | 120 h @ 180 mA/cm² | Nat. Comm., 2024 |
Key Observation: A pronounced decline in FE(C₂H₄) and energy efficiency is consistently observed when moving from optimized lab-scale half-cells (H-cell) to integrated membrane electrode assemblies (MEAs) and further to multi-cell stacks. This is attributed to mass transport limitations, ohmic losses, reactant crossover, and current/voltage distribution inefficiencies at scale.
Experimental Protocols for Benchmarking
To ensure comparable data, the following standardized protocols are recommended based on consensus in recent literature.
Protocol 1: Lab-Scale Catalyst Activity & FE Testing (H-Cell)
- Electrode Preparation: Catalyst ink (5 mg catalyst, 950 µL isopropanol, 50 µL Nafion binder) is sonicated for 60 min and drop-cast onto a carbon paper gas diffusion layer (GDL) to achieve 1 mg/cm² loading.
- Cell Setup: Use a two-compartment H-cell separated by a Nafion 117 membrane. The cathode chamber is filled with 30 mL of 0.1 M KHCO₃ electrolyte, saturated with CO₂ via bubbling for 30 min prior to and during the experiment.
- Electrolysis: Perform chronoamperometry at fixed potentials (vs. RHE) using a potentiostat. CO₂ is continuously fed at 20 sccm.
- Product Analysis: Gaseous effluent is analyzed online via a gas chromatograph (GC) equipped with a flame ionization detector (FID) and a thermal conductivity detector (TCD). Liquid products are quantified using NMR.
- FE Calculation: FE = (z * n * F) / Q, where z is the number of electrons transferred (12 for C₂H₄), n is moles of product, F is Faraday's constant, and Q is total charge passed.
Protocol 2: MEA Cell & Stack Performance Evaluation
- MEA Fabrication: Catalyst-coated membrane (CCM) method is used. Cathode catalyst ink is spray-coated directly onto an anion exchange membrane (AEM) to form a 2 cm² active area with 2 mg/cm² loading. The anode (IrO₂) is similarly applied.
- Single Cell Assembly: The CCM is sandwiched between two PTFE-coated carbon GDLs and compressed in a flow field plate (graphite or titanium) at 2 N·m torque.
- Stack Assembly: 5-10 identical MEAs are assembled with bipolar plates featuring integrated flow fields. Uniform compression is critical (typically 1.5-2 N·m per bolt).
- System Operation: CO₂ is humidified and fed to the cathode at 100 sccm/cm². Anolyte (0.1 M KOH) is circulated. Cell temperature is maintained at 60°C.
- Performance Metrics: Voltage of each cell is monitored individually. Product analysis is performed on the combined cathode outlet stream via GC. Stability tests are run at constant current.
Visualizing Scalability Challenges and Workflows
Diagram Title: Hurdles in Scaling CO2 to Ethylene Conversion Systems
Diagram Title: Hierarchical Experimental Workflow for Scalability Testing
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for CO₂-to-Ethylene Scalability Research
| Item | Function & Relevance for Scalability |
|---|---|
| Anion Exchange Membrane (AEM) | Critical for MEA and stack design. Conducts hydroxide ions, determines CO₂ crossover rate, and impacts cathode local environment. Example: Sustainion, PiperION. |
| Gas Diffusion Layer (GDL) | PTFE-coated carbon paper or felt. Provides structural support, gas/electron transport, and product removal. Hydrophobicity management is key for flooding prevention at high current density. |
| Copper-Based Catalyst Inks | Precursors for electrode fabrication. Include catalyst nanoparticles, ionomer (e.g., Sustainion anionomer), and solvent. Dispersion quality dictates catalyst layer morphology and activity. |
| Bipolar Plates (Graphite/Ti-coated) | Used in stacks to distribute current, gases, and coolant between cells. Must be corrosion-resistant and have high electrical conductivity. |
| High-Precision Potentiostat/Galvanostat | For accurate multi-channel electrochemical measurement, essential for individual cell monitoring in a stack and long-term stability tests. |
| Micro-Gas Chromatograph (µ-GC) | Enables rapid, online analysis of gaseous products (C₂H₄, CO, CH₄, H₂) from both single-cell and stack outlets, crucial for real-time FE calculation. |
| Reference Electrode (e.g., Ag/AgCl) | Required for half-cell testing to measure true catalyst potential vs. RHE, establishing baseline performance before MEA integration. |
| In-situ Raman/FTIR Cell | For diagnosing reaction intermediates and catalyst state under operating conditions, helping identify degradation pathways during scale-up. |
Benchmarking Performance: A Critical Review of State-of-the-Art Systems and Catalysts
This guide, framed within a broader thesis on Faraday efficiency (FE) benchmarks for CO₂-to-ethylene (C₂H₄) conversion, provides an objective comparison of recent high-performance electrocatalysts. The electrochemical reduction of CO₂ to multi-carbon products, especially ethylene, is a critical pathway for renewable chemical synthesis and energy storage. This analysis focuses on three key performance metrics: C₂H₄ Faradaic Efficiency (FE), Current Density (Rate), and Operational Stability.
Key Performance Data Comparison
The following table summarizes quantitative data for recent record-holding catalysts as reported in premier scientific literature (data sourced from Nature, Science, JACS, and Nature Energy, 2023-2024).
| Catalyst System & Citation | Max C₂H₄ FE (%) | Partial Current Density (mA cm⁻²) | Total Current Density (mA cm⁻²) | Stability (Hours) | Key Electrolyte | Overpotential (vs. RHE) |
|---|---|---|---|---|---|---|
| Cu-Ag Tandem Dendrites (Nat. Energy, 2024) | 75 | 500 | >800 | 150 | 1 M KOH | ~0.9 V |
| OMA-Cu (Ordered Macroporous) (Science, 2023) | 72 | 400 | 550 | 100 | 1 M KOH | ~0.85 V |
| Polyamine-Modified Cu Oxide (Nat. Catal., 2023) | 70 | 300 | 430 | 200 | 0.1 M KHCO₃ | ~0.95 V |
| Cu-PDI Molecular Hybrid (JACS, 2024) | 68 | 250 | 370 | 80 | 1 M KOH | ~0.88 V |
| Nanoporous Cu₂S-Derived Cu (Nat. Commun., 2023) | 66 | 600 | 900 | 50 | 7 M KOH | ~0.8 V |
Detailed Experimental Protocols
General H-Cell Electrolysis Protocol (Baseline)
- Electrode Preparation: Catalyst ink is prepared by dispersing 5 mg of catalyst powder in 1 mL of solution (950 µL isopropanol + 50 µL 5 wt% Nafion). The ink is sonicated for 60 min, then uniformly coated onto a gas diffusion layer (GDL) or carbon paper (1x1 cm²) to achieve a loading of ~0.5 mg cm⁻².
- Cell Assembly: A standard H-cell separated by a Nafion 117 membrane is used. The catalyst-coated GDL serves as the working electrode. A Pt wire and an Ag/AgCl (saturated KCl) electrode are used as counter and reference electrodes, respectively.
- Electrolyte & Conditions: 30 mL of electrolyte (e.g., 1 M KOH) is purged with CO₂ for 30 min prior to experiment. During electrolysis, CO₂ is continuously fed into the cathode compartment at a rate of 20 sccm.
- Electrolysis: Potentiostatic electrolysis is performed using a potentiostat (e.g., BioLogic SP-300) at the specified potential (converted to RHE scale).
- Product Analysis:
- Gas Products: Analyzed via online gas chromatography (GC, e.g., Agilent 8890) equipped with both TCD and FID detectors, using Ar/CH₄ as carrier gas. Quantification is via external calibration curves.
- Liquid Products: Analyzed by NMR spectroscopy (e.g., ¹H NMR on a 600 MHz spectrometer) using dimethyl sulfoxide (DMSO) as an internal standard.
Flow Cell Protocol (for High Current Density Tests)
- Membrane Electrode Assembly (MEA): The catalyst layer is coated directly onto the microporous layer of a PTFE-treated carbon GDL. This cathode and a Ni foam anode are assembled with an anion exchange membrane (e.g., Sustainion) in a commercial flow cell.
- Electrolyte Circulation: 1 M KOH is circulated on the cathode side (CO₂-saturated) at 10 mL min⁻¹ using a peristaltic pump. Anolyte (1 M KOH) is circulated separately.
- Operation: High-purity CO₂ is fed to the cathode gas chamber at a controlled flow rate. Electrolysis is performed at constant current.
- Stability Test: The cell is operated continuously at a fixed current density, with periodic sampling of gas and liquid outlets for product analysis to monitor performance decay.
Visualizing Catalyst Performance & Pathways
Diagram Title: Pathways and Competing Reactions in CO2-to-C2H4 Conversion
Diagram Title: Experimental Workflow for Catalyst Benchmarking
The Scientist's Toolkit: Research Reagent Solutions
Essential materials and reagents critical for conducting and reproducing high-level CO₂-to-C₂H₄ electrocatalysis research.
| Item | Function & Relevance |
|---|---|
| Gas Diffusion Layer (GDL) (e.g., Sigracet 39BB) | Porous carbon substrate for catalyst loading. Ensures efficient triple-phase (CO₂ gas/electrolyte/catalyst) contact, crucial for high current densities. |
| Anion Exchange Membrane (e.g., Sustainion X37-50) | Separates cathode and anode compartments in flow cells while allowing hydroxide ion transport. Critical for stable operation in alkaline media. |
| High-Purity CO₂ (≥ 99.999%) with Isotopic ¹³CO₂ | Reactant gas. ¹³C-labeled CO₂ is essential for confirming the carbon source of products via NMR or mass spectrometry. |
| Potentiostat/Galvanostat (e.g., BioLogic VSP-300) | Precision instrument for applying potential/current and measuring electrochemical response. Essential for controlled electrolysis and in-situ techniques. |
| Online Gas Chromatograph (e.g., Agilent 8890 GC with TCD/FID) | For real-time, quantitative analysis of gaseous products (H₂, CO, C₂H₄, CH₄, etc.). The primary tool for calculating Faradaic efficiency. |
| NMR Solvent with Internal Standard (D₂O, DMSO-d6) | Solvent for ¹H NMR analysis of liquid products (e.g., ethanol, acetate). An internal standard (e.g., DMSO) enables precise quantification. |
| Customized Flow Cell Reactor (e.g., Dioxide Materials or custom design) | Enables testing under industrially relevant high current densities (> 200 mA cm⁻²) by addressing mass transport limitations of CO₂. |
This comparison guide analyzes the performance of single-crystal and nanostructured copper catalysts for the electrochemical reduction of CO₂ to ethylene (C₂H₄), a critical reaction for sustainable chemical synthesis. The evaluation is framed within the broader thesis of achieving high Faraday efficiency (FE) benchmarks for this conversion.
Performance Comparison & Experimental Data
The catalytic performance is governed by a fundamental trade-off: single-crystal Cu offers precise, tunable selectivity, while nanostructured Cu provides higher intrinsic activity due to its abundance of active sites.
Table 1: Comparative Performance Metrics for CO₂ to C₂H₄ Conversion
| Catalyst Type | Typical FE for C₂H₄ (%) | Partial Current Density (mA/cm²) | Onset Potential (vs. RHE) | Key Structural Feature | Stability Notes |
|---|---|---|---|---|---|
| Single-Crystal Cu(100) | 40-60 | 5-15 | ~ -0.9 V | Well-defined (100) terraces | High structural stability; deactivation mainly from impurities. |
| Single-Crystal Cu(111) | < 10 | 1-5 | ~ -1.1 V | Close-packed (111) facets | Stable, but primarily produces CH₄ or H₂. |
| Nanostructured Cu (e.g., nanoparticles) | 50-70 | 15-30 | ~ -0.7 V | High density of grain boundaries, edges, steps | Activity can decay due to aggregation or reconstruction. |
| Oxide-Derived Nanostructured Cu | 60-75 | 20-50 | ~ -0.6 V | Residual oxygen, complex porous morphology | Highly active but may undergo gradual reduction. |
Table 2: Key Reaction Intermediates and Proposed Selectivity Determinants
| Catalyst Type | Proposed Key Intermediate for C₂H₄ | Competing Pathway | Reaction Condition Influence |
|---|---|---|---|
| Single-Crystal Cu(100) | CO-CO dimerization at terrace sites | *CO protonation to CH₄ on steps/defects | Highly sensitive to electrolyte pH (alkaline favored). |
| Nanostructured Cu | *CO dimerization at grain boundaries | Hydrogen Evolution Reaction (HER) | Local pH and mass transport effects are significant. |
Experimental Protocols for Key Studies
1. Protocol for Single-Crystal Cu(100) Electrochemical Testing:
- Catalyst Preparation: A Cu(100) single-crystal disk is oriented, cut, and polished mechanically and electrochemically. It is then annealed in a hydrogen flame and quenched in ultrapure water under an inert atmosphere to preserve a clean, ordered surface.
- Electrochemical Cell: A three-electrode H-cell separated by an ion-exchange membrane.
- Electrolyte: 0.1 M KHCO₃ or KOH solution, saturated with CO₂.
- Procedure: The crystal is transferred to the cell without air exposure. Cyclic voltammetry is performed, followed by chronoamperometry at fixed potentials (typically -0.8 to -1.2 V vs. RHE). Gas and liquid products are quantified via online gas chromatography and NMR, respectively.
2. Protocol for Oxide-Derived Nanostructured Cu Synthesis & Testing:
- Synthesis: A Cu foil is anodized or thermally oxidized in air to form a CuO/Cu₂O layer.
- Electrochemical Pre-treatment: The oxidized foil is used as a cathode in a CO₂-saturated electrolyte. A potentiodynamic reduction protocol (e.g., cycling from 0 to -1.2 V vs. RHE) reduces the oxide in situ, creating a metastable, nanostructured Cu surface.
- Performance Testing: Following stabilization, chronoamperometry is conducted at target potentials. Product analysis is performed as above. Operando techniques like XAS may be used to monitor Cu oxidation state.
Visualizing the Activity-Selectivity Trade-off
Diagram 1: The Core Catalytic Design Trade-off
Diagram 2: Key Reaction Pathways from *CO Intermediate
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials and Reagents for CO₂-to-C₂H₄ Catalyst Research
| Item | Function & Rationale |
|---|---|
| Single-Crystal Cu Electrodes (e.g., (100), (111)) | Provide atomically defined surfaces to establish fundamental structure-activity relationships and baseline selectivity. |
| High-Purity CO₂ Gas (≥ 99.999%) | Minimizes contamination from sulfur or other compounds that poison Cu catalysts, ensuring reproducible activity. |
| Isotope-Labeled ¹³CO₂ | Allows for precise tracking of carbon atoms in products via Mass Spectrometry or NMR, confirming the product origin. |
| Deuterated Water (D₂O) / Deuterated Electrolytes | Used in mechanistic studies to probe proton-coupled electron transfer steps and kinetic isotope effects. |
| Ion-Exchange Membranes (Nafion, Sustainion, etc.) | Separates anodic and cathodic chambers, controlling ion transport (H⁺, K⁺, HCO₃⁻) which critically impacts local pH and reaction selectivity. |
| Online Gas Chromatography (GC) System | Equipped with FID and TCD detectors for quantitative, real-time analysis of gaseous products (C₂H₄, CH₄, CO, H₂). |
| ¹H-NMR Spectroscopy | Essential for quantifying liquid-phase multi-carbon products (e.g., ethanol, n-propanol) using an internal standard (e.g., DMSO). |
| In-situ/Operando Cell for X-ray Absorption Spectroscopy (XAS) | Enables monitoring of the Cu oxidation state and local coordination environment under actual reaction conditions. |
Electrochemical CO₂ reduction (CO2R) to ethylene (C₂H₄) is a critical pathway for sustainable chemical production. Two dominant reactor architectures are the Flow Cell and the Membrane Electrode Assembly (MEA). This guide objectively compares their performance in balancing the critical triad of Faraday Efficiency (FE), Energy Efficiency (EE), and Operational Durability within CO2R research.
Performance Comparison: Key Metrics
The following table summarizes benchmark performance data from recent high-impact studies (2022-2024).
Table 1: Performance Comparison of Flow Cell vs. MEA for CO₂ to Ethylene
| Metric | Flow Cell (Alkaline) | MEA (Zero-Gap) | Notes & Conditions |
|---|---|---|---|
| Max C₂H₄ FE (%) | 65 - 75% | 50 - 65% | Flow cells typically achieve higher single-pass FEs due to optimized local pH. |
| Full Cell EE (%) | 20 - 30% | 30 - 40% | MEAs often show superior EE due to lower ohmic losses from zero-gap design. |
| Current Density (mA/cm²) | 200 - 800 | 200 - 500 | Flow cells enable higher current densities via enhanced mass transport. |
| C₂H₄ Partial Current (mA/cm²) | ~150 - 500 | ~100 - 300 | Direct indicator of production rate. |
| Stability (Hours) | 100 - 1000+ | 50 - 200 | MEAs face greater challenges with cation crossover and membrane degradation. |
| Key Advantage | High FE, High Rate | High EE, Simpler System | |
| Primary Limitation | Carbonate Formation, Liquid Management | Crossover, Dehydration, Catalyst Degradation |
Experimental Protocols for Benchmarking
To generate comparable data, standardized protocols are essential.
Protocol 1: Flow Cell Testing (Gas Diffusion Electrode)
- Cell Assembly: A typical H-cell or microfluidic flow cell is used. The cathode is a Cu-based Gas Diffusion Electrode (GDE) pressed against a polytetrafluoroethylene (PTFE) or polycarbonate flow field.
- Electrolyte: 1-5 M KOH or KHCO₃ solution is circulated past the back of the GDE at 5-20 mL/min. The anode compartment uses the same electrolyte.
- Gas Supply: High-purity CO₂ (≥ 99.999%) is fed to the GDE's gas chamber at a controlled rate (10-50 sccm).
- Electrolysis: Performed using a potentiostat/galvanostat. Potentials are reported versus the Reversible Hydrogen Electrode (RHE).
- Product Analysis: Gas effluent is directed to a Gas Chromatograph (GC) equipped with FID and TCD detectors for quantification every 15-30 mins. Liquid products are analyzed via NMR or HPLC.
Protocol 2: MEA Cell Testing
- MEA Fabrication: A cathode GDE (Cu catalyst) and an anode (IrO₂/Pt for OER) are hot-pressed onto either side of an anion exchange membrane (AEM).
- Cell Assembly: The MEA is clamped between two graphite or titanium flow fields in a zero-gap configuration.
- Reactant Supply: Humidified CO₂ is fed to the cathode chamber. Liquid water or humidified gas is supplied to the anode.
- Electrolysis & Analysis: Similar potentiostatic control as Protocol 1. Gas products from both outlets are analyzed by online GC. Water from the anode is collected for liquid product analysis.
System Architecture & Performance Trade-offs
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for CO2R Reactor Studies
| Item | Function | Example Product/Chemical |
|---|---|---|
| Anion Exchange Membrane | Conducts hydroxide ions, separates compartments while allowing ion flow. Critical for MEA stability. | Sustainion X37-50, Fumasep FAA-3, Tokuyama A201 |
| Gas Diffusion Layer (GDL) | Provides structural support, gas/liquid pathways, and electron conduction for the catalyst. | Sigracet 39BB, Freudenberg H23, Toray Carbon Paper |
| Copper Nanoparticle Catalyst | The active material for multi-carbon product formation (e.g., C₂H₄). | Cu NPs on Carbon, Oxide-Derived Cu, Cu Nanocubes |
| Alkaline Electrolyte | Creates high-pH environment at cathode to suppress HER and favor C-C coupling. | Potassium Hydroxide (KOH), Potassium Bicarbonate (KHCO₃) |
| Ionomer Binder | Binds catalyst particles to GDL and creates ion-conductive network within the catalyst layer. | Sustainion ionomer, PiperION, Nafion (for certain configurations) |
| Reference Electrode | Provides stable potential reference to accurately report cathode potential vs. RHE. | Reversible Hydrogen Electrode (RHE), Double Junction Ag/AgCl |
Critical Performance Pathways
Accurate validation of electrochemical CO₂ reduction (CO2R) performance, specifically for ethylene (C₂H₄) production, hinges on standardized reporting protocols and rigorous product detection. Inconsistent methodology undermines the comparability of Faraday efficiency (FE) benchmarks critical for advancing this research. This guide compares performance claims for key catalyst systems, emphasizing the experimental protocols that substantiate them.
Comparative Performance of Select CO2R-to-Ethylene Catalysts
Data sourced from recent literature (2023-2024). Performance is highly dependent on exact conditions and cell design.
| Catalyst System | Typical Operating Potential (vs. RHE) | Reported FE(C₂H₄) (%) | Stability Duration | Key Differentiating Protocol Detail |
|---|---|---|---|---|
| Oxide-Derived Copper (OD-Cu) | -0.9 V | ~55% | 10 hours | Online GC calibrated with in-line IR detector for carbon balance. |
| Polycrystalline Copper | -1.1 V | <30% | < 2 hours | ¹³C isotopic labeling to confirm ethylene carbon source. |
| Bimetallic Cu-Ag Nanostructures | -0.8 V | 65% | 20 hours | NMR for liquid products; post-operation SEM for morphology correlation. |
| Molecular Cu-Complex / GDE | -0.7 V | 42% | 5 hours | Rigorous protocol for quantifying dissolved gases via headspace GC. |
Detailed Experimental Methodologies
1. Online Gas Chromatography (GC) with Carbon Balance
- Purpose: Quantify gaseous products (C₂H₄, CH₄, CO, H₂) and calculate Faradaic efficiencies.
- Protocol: a. Electrolyte is pre-saturated with CO₂ and continuously sparged during operation. b. Effluent gas stream is directed to a gas chromatograph via a heated transfer line. c. GC is equipped with both Thermal Conductivity (TCD) and Flame Ionization (FID) detectors. d. Critical Step: A known flow of an internal standard gas (e.g., 1% Ar in CO₂) is introduced pre-reactor. Comparing inlet/outlet Ar concentrations allows for precise calculation of total gas outflow and carbon balance (>95% required for valid FE).
2. ¹³C Isotopic Labeling and Nuclear Magnetic Resonance (NMR)
- Purpose: Unambiguously identify the carbon source in products and quantify liquid-phase products.
- Protocol: a. Experiments are performed using 99% ¹³CO₂ as the feed gas. b. Gaseous products are analyzed by GC-MS to confirm mass shift (e.g., ¹³C₂H₄ vs. ¹²C₂H₄). c. Liquid electrolyte is periodically sampled and analyzed by ¹H and ¹³C NMR using a water suppression pulse sequence. Quantification is achieved via an internal standard (e.g., dimethyl sulfoxide).
3. Post-Mortem Catalyst Characterization
- Purpose: Link performance claims to catalyst structure and stability.
- Protocol: a. Post-electrolysis, the electrode is rinsed thoroughly with deionized water and dried under N₂. b. Scanning Electron Microscopy (SEM) is performed to assess morphology changes (e.g., reconstruction, agglomeration). c. X-ray Photoelectron Spectroscopy (XPS) is conducted ex situ to analyze surface composition and oxidation state, noting potential air exposure artifacts.
Experimental Workflow for Validated FE Claims
Diagram Title: Workflow for Validating CO2R-to-Ethylene Performance
The Scientist's Toolkit: Key Research Reagent Solutions
| Item | Function in CO2R-to-Ethylene Research |
|---|---|
| ¹³C-Labeled CO₂ (99%) | Isotopic tracer to confirm carbon source in products, eliminating contamination artifacts. |
| High-Purity Electrolyte Salts (e.g., KHCO₃, KClO₄) | Minimizes impurity-driven side reactions and ensures reproducible ionic strength. |
| Internal Standard Gases (e.g., 1% Ar/CO₂, 1% CH₄/He) | Enables accurate quantification of gas flow rates and GC detector response factors. |
| NMR Internal Standards (e.g., DMSO-d6, DSS) | Provides a reference for chemical shift and enables quantitative analysis of liquid products. |
| Ion-Exchange Membranes (e.g., Nafion 117, Sustainion) | Separates anolyte and catholyte, controlling pH and cross-over in membrane electrode assemblies. |
| Gas Diffusion Layer (GDL) Electrodes | Provides high CO₂ flux in flow cells, enabling industrially relevant current densities. |
Within the context of establishing Faraday efficiency (FE) benchmarks for CO₂-to-ethylene conversion research, this comparison guide evaluates the performance requirements for economically viable electrochemical systems. The analysis is grounded in current Techno-Economic Analysis (TEA) studies that define the necessary operational parameters for cost-competitive green ethylene production.
TEA-Derived Performance Targets for CO₂-to-Ethylene Electrolysis
Recent TEA models converge on key performance metrics that an electrolyzer must achieve to compete with steam cracking of fossil fuels. The consensus indicates that both high selectivity (Faraday Efficiency) and long-term stability are non-negotiable for reducing the levelized cost of ethylene (LCOE).
Table 1: Summary of TEA-Derived Performance Targets for Cost-Competitive Ethylene Production
| Performance Parameter | Baseline Performance (Current State) | TEA Target for Competitiveness | Stretch Target (Advanced Scenarios) |
|---|---|---|---|
| Faraday Efficiency to C₂H₄ (%) | 60-75% (in lab-scale H-cells) | ≥ 85% | ≥ 90% |
| Full-Cell Energy Efficiency (%) | ~30-40% | ≥ 50% | ≥ 60% |
| Current Density (mA/cm²) | 100-300 (at high FE) | 300-500 (maintaining high FE) | > 1000 |
| Catalyst/System Stability | < 100 hours (continuous) | ≥ 1,000 hours | ≥ 10,000 hours |
| Single-Pass Carbon Conversion (%) | < 10% (flow cells) | ≥ 30% | ≥ 50% |
| Projected Ethylene Cost ($/ton) | > 2000 | ≤ 1,200 - 1,400 (fossil parity) | < 1000 |
The data indicates that an FE of at least 85% is critical, but not sufficient alone. This high FE must be sustained at industrially relevant current densities (>300 mA/cm²) for thousands of hours to amortize capital costs.
Comparison of Catalytic Systems Against TEA Targets
Experimental data from recent high-profile studies are compared against the TEA benchmarks.
Table 2: Experimental Performance of Recent CO₂-to-C₂H₄ Electrocatalysts
| Catalyst System & Cell Type | Max C₂H₄ FE (%) | Current Density (mA/cm²) | Stability Duration | Key Limitation vs. TEA Target |
|---|---|---|---|---|
| Polycrystalline Cu (H-cell) | ~55% | ~10 | N/A | Low current density, moderate FE. |
| Oxide-Derived Cu Nanowires (Flow Cell) | 70% | 300 | 150 h | FE below target; stability limited. |
| Cu-Ag Tandem Catalyst (MEA) | 80% | 500 | 100 h | FE near target, but stability far short. |
| MOF-Derived Cu Catalyst (Flow Cell) | 87% | 350 | 200 h | Meets FE target, stability insufficient. |
| Alkaline Membrane Electrode (MEA) | 85% | 750 | 190 h | Meets FE & current density, stability insufficient. |
| Cu-Polymer Hybrid System (Flow Cell) | 82% | 250 | 1000 h | Meets stability benchmark, FE marginally low. |
Experimental Protocols for Key Cited Studies
Protocol 1: Flow Cell Testing for Cu Nanowire Catalyst
- Electrode Preparation: Anodic IrO₂ on Ti felt. Cathodic Gas Diffusion Electrode (GDE) is prepared by spray-coating a catalyst ink (oxide-derived Cu nanowires, Nafion binder, isopropanol) onto carbon paper.
- Electrolyte: 1 M KOH aqueous solution circulated on the cathode side at 10 mL/min. Anolyte is 1 M KHCO₃.
- Electrolysis: Performed in a two-compartment flow cell separated by an anion exchange membrane. CO₂ is fed to the cathode chamber at 20 sccm.
- Product Analysis: Gaseous effluent analyzed by online GC (TCD for H₂, CO; FID for hydrocarbons). Liquid products quantified via NMR.
- Stability Testing: Potentiostatic hold at -0.7 V vs. RHE; performance metrics logged every hour.
Protocol 2: Membrane Electrode Assembly (MEA) Testing
- MEA Fabrication: Cathode catalyst (e.g., Cu-Ag) is directly coated onto a porous carbon layer of the gas diffusion layer. This is hot-pressed against an anion exchange membrane with an anodic catalyst layer.
- Cell Assembly: The MEA is sandwiched between two graphite flow plates with serpentine channels.
- Operation: Humidified CO₂ is fed to the cathode; water or humidified gas is fed to the anode. No liquid electrolyte is used.
- Analysis: Similar online GC analysis. High current density is enabled by zero-gap configuration and improved mass transport.
Visualizations
Title: Interdependence of Performance Metrics for TEA Targets
Title: Standard Workflow for CO2 to Ethylene Performance Testing
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for CO₂-to-C₂H₄ Electrocatalysis Research
| Reagent/Material | Function & Rationale | Common Example |
|---|---|---|
| Copper-based Precursors | Source for electrocatalyst synthesis. Morphology and oxidation state are key to C-C coupling. | Copper nitrate, copper sulfate, copper foil, Cu₂O. |
| Gas Diffusion Layer (GDL) | Porous, conductive substrate for cathode in flow cells/MEA. Enables high current density by improving CO₂ transport. | Carbon paper (e.g., Sigracet), carbon cloth. |
| Ion Exchange Membranes | Separates anode and cathode compartments, selectively transports ions (H⁺, OH⁻, HCO₃⁻). | Nafion (cation exchange), Sustainion (anion exchange). |
| High-Purity CO₂ Gas | Reactant feed. Impurities (e.g., O₂) can alter catalyst performance. | 99.999% CO₂, often with in-line purification filters. |
| Electrolyte Salts | Provide ionic conductivity and influence local pH, which critically affects reaction pathways. | Potassium hydroxide (KOH), potassium bicarbonate (KHCO₃). |
| Internal Standard for GC | Allows for quantitative calibration of gas product streams during online analysis. | Argon (Ar) or Neon (Ne), mixed with effluent. |
| Deuterated Solvent for NMR | For quantitative analysis of liquid-phase products (e.g., ethanol, acetate). | D₂O with a known concentration of internal standard (e.g., DMSO). |
Conclusion
Faraday Efficiency remains the pivotal benchmark for assessing the viability of electrochemical CO2-to-ethylene conversion. Progress hinges on an integrated approach, combining tailored catalyst design, optimized electrolyte and reactor engineering, and robust diagnostic protocols to mitigate parasitic reactions. While recent breakthroughs in copper-based catalysts and flow cell configurations have pushed FE benchmarks beyond 70% in some studies, challenges in long-term stability, scalability, and energy efficiency persist. The future trajectory for researchers involves developing dynamic operation strategies, exploring beyond-copper catalysts, and creating standardized testing protocols to accurately compare systems. Success in this field promises not only a sustainable carbon-negative route to a foundational chemical but also a platform technology adaptable for synthesizing other valuable multi-carbon products critical to the chemical and pharmaceutical industries.