TPD vs. Isothermal Chemisorption: A Comparative Guide to Catalyst Site Characterization for Drug Development
This article provides a comprehensive guide for researchers and drug development professionals on the strategic use of Temperature-Programmed Desorption (TPD) and Isothermal Chemisorption for catalyst site characterization.
TPD vs. Isothermal Chemisorption: A Comparative Guide to Catalyst Site Characterization for Drug Development
Abstract
This article provides a comprehensive guide for researchers and drug development professionals on the strategic use of Temperature-Programmed Desorption (TPD) and Isothermal Chemisorption for catalyst site characterization. We explore the fundamental principles of each technique, detailing their specific methodologies and applications in measuring active site density and strength. The guide addresses common experimental challenges, offers optimization strategies, and presents a direct comparison of the data each method yields. By synthesizing this information, we empower scientists to select the optimal technique for their specific material and research question, ultimately enhancing catalyst design for pharmaceutical synthesis and biomedical applications.
Core Principles of Active Site Analysis: Understanding TPD and Chemisorption Fundamentals
The efficiency and selectivity of catalytic processes in drug synthesis are dictated by the nature of the active site. A core thesis in modern heterogeneous catalysis research is that Temperature-Programmed Desorption (TPD) and isothermal chemisorption offer complementary, yet distinct, insights into active site characterization. This guide compares these two pivotal techniques within the context of catalytic drug synthesis.
Comparative Analysis: TPD vs. Isothermal Chemisorption
The following table summarizes the key performance characteristics of TPD and isothermal chemisorption for active site analysis.
Table 1: Technique Comparison for Active Site Characterization
| Feature | Temperature-Programmed Desorption (TPD) | Isothermal Chemisorption |
|---|---|---|
| Primary Information | Energetic distribution & strength of adsorbate binding; site heterogeneity. | Quantitative number of accessible sites; active site density under reaction conditions. |
| Typical Data Output | Desorption rate vs. temperature; peak temperatures (Tp) indicate binding energy. | Uptake (molecules/g) vs. pressure at constant temperature; isotherm fit to Langmuir or other models. |
| Operational Mode | Dynamic, non-isothermal. Temperature is linearly increased. | Static or flow, isothermal. Measurement occurs at a fixed, controlled temperature. |
| Probe Flexibility | High. Can use NH3 (acidity), CO2 (basicity), H2 (metal sites), specific drug intermediates. | Moderate. Requires careful selection of probe molecule and temperature to avoid multilayer formation or reaction. |
| Strength vs. Number | Excellent for determining strength and heterogeneity of sites. | Excellent for determining number of accessible sites. |
| Throughput | Moderate; requires careful temperature programming. | Can be higher for single-point measurements, but full isotherms are time-intensive. |
| Key Limitation | May alter or sinter sensitive sites during heating. Indirect quantification. | Assumes a known stoichiometry for adsorption; may miss weak or very strong sites. |
Table 2: Experimental Data from a Model Pd/C Catalyst for Suzuki-Miyaura Cross-Coupling*
| Catalyst | TPD-H2 Peak Max (ºC) | Isothermal H2 Chemisorption (μmol/g) | Calculated Pd Dispersion (%) | Suzuki Reaction Yield (%) |
|---|---|---|---|---|
| Pd/C (Low Dispersion) | 125, 285 | 45 | 12 | 78 |
| Pd/C (High Dispersion) | 85, 250 | 180 | 48 | 95 |
| Sintered Pd/C | 290 | 22 | 6 | 35 |
*Synthetic data representative of typical literature trends.
Experimental Protocols
Protocol 1: Temperature-Programmed Desorption (TPD) of Probe Molecules
- Pretreatment: Load 50-100 mg of catalyst into a U-shaped quartz tube. Purge with inert gas (He, Ar) at 300°C for 1-2 hours to clean the surface.
- Adsorption: Cool to adsorption temperature (e.g., 50°C for NH3, 35°C for H2). Expose to a calibrated pulse or flow of pure probe gas until saturation is achieved.
- Purge: Switch to inert carrier gas at the adsorption temperature to remove physisorbed species (typically 30-60 mins).
- Desorption: Heat the sample linearly (e.g., 10°C/min) to a final temperature (e.g., 600°C) under the inert carrier gas flow.
- Detection: Monitor desorbing species using a thermal conductivity detector (TCD) or mass spectrometer (MS). Calibrate the detector response with known volumes of the probe gas.
Protocol 2: Static Volumetric Isothermal Chemisorption
- System Preparation: Evacuate the sample manifold and sample cell to ultra-high vacuum (<10-5 Torr). Pre-treat the catalyst sample in-situ (e.g., H2 reduction at 200°C, followed by evacuation).
- Dosing: Isolate the sample cell and introduce a small, known dose of adsorbate gas (e.g., H2, CO) from a calibrated volume. Allow the system to reach equilibrium (pressure stabilizes).
- Measurement: Record the equilibrium pressure. The amount adsorbed is calculated from the pressure drop using the ideal gas law.
- Iteration: Repeat steps 2-3 to generate an adsorption isotherm across a range of pressures.
- Analysis: Extrapolate the linear portion of the isotherm to zero pressure to determine the strong chemisorption uptake. Calculate metal dispersion assuming a stoichiometry (e.g., H:Pt = 1:1, CO:Pt = 1:1).
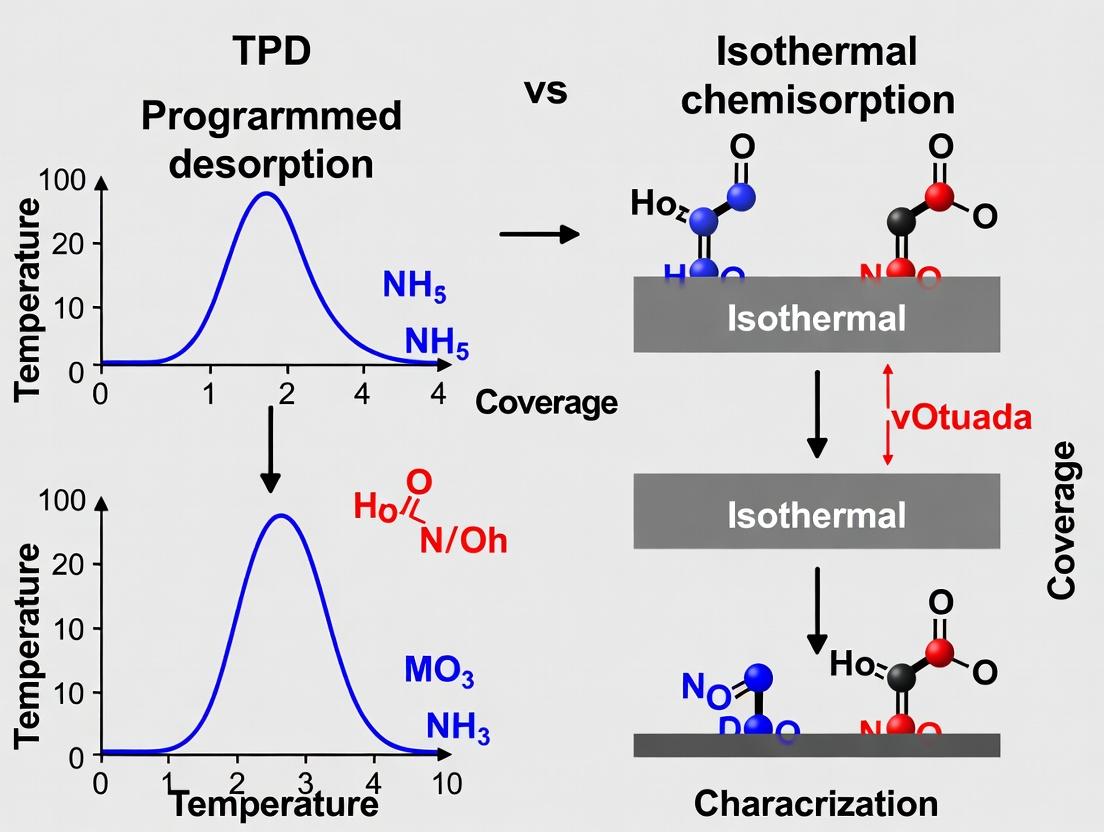
Visualizing the Characterization Workflow
Title: Complementary Data Pathways from TPD and Chemisorption
Title: Experimental Workflow for Surface Site Characterization
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Active Site Characterization Studies
| Item | Function in Characterization |
|---|---|
| High-Purity Probe Gases (e.g., 5% H2/Ar, 10% NH3/He, 99.999% CO) | Serve as molecular probes to titrate specific active sites (metal, acid, base). Purity is critical to avoid site poisoning. |
| Calibrated Micrometering Valves & Mass Flow Controllers (MFCs) | Precisely control the flow rate of carrier and probe gases during TPD or pulse chemisorption experiments. |
| Thermal Conductivity Detector (TCD) or Mass Spectrometer (MS) | TCD is standard for quantifying desorption; MS is essential for identifying desorbing species and detecting fragments. |
| High-Vacuum System & Pressure Transducers | Required for static volumetric chemisorption. Allows precise measurement of minute pressure changes for uptake calculation. |
| Quartz U-Tube Microreactor | Standard sample holder for TPD studies. Inert, can withstand high temperatures, and allows for in-situ pretreatment. |
| Reference Catalysts (e.g., EUROPT-1, standard Al2O3) | Well-characterized materials with known metal dispersion or acid site density used to validate instrument performance and methodology. |
Comparative Analysis: TPD versus Isothermal Chemisorption for Site Characterization
Surface characterization of catalysts and pharmaceutical adsorbents is critical for understanding performance. This guide objectively compares two principal techniques: Temperature-Programmed Desorption (TPD) and Isothermal Chemisorption, within a thesis focused on elucidating active site energetics and distribution.
Performance Comparison Table
| Feature / Metric | Temperature-Programmed Desorption (TPD) | Isothermal Chemisorption (Pulse or Flow) |
|---|---|---|
| Primary Information | Desorption energetics (Ed), activation energy, binding site heterogeneity, surface coverage. | Total active site density (μmol/g), dispersion (%), average particle size. |
| Kinetic Resolution | High. Can deconvolute multiple binding states with different energies via heating ramps. | Low. Provides an integral, coverage-averaged measurement at a single temperature. |
| Experimental Temperature | Dynamic range (often 300-1200 K). | Single, isothermal point (often 300-500 K). |
| Probe Flexibility | High. Any desorbing gas can be monitored (NH₃, CO₂, H₂, O₂, etc.). | Moderate. Limited to strong, irreversible chemisorption at chosen temperature. |
| Quantification Method | Integration of desorption peaks. Requires calibration and careful baseline subtraction. | Direct volumetric/titration measurement of irreversibly held gas. |
| Typical Data Output | Spectra (rate vs. T) showing peaks corresponding to distinct site energies. | Single numerical value for uptake (μmol/g). |
| Key Strength | Reveals energetic landscape and heterogeneity of sites. | Simpler, faster, more directly quantitative for total site count. |
| Main Limitation | Quantitative accuracy can be compromised by re-adsorption, readsorption, and heating rate effects. | Assumes uniform site energy; misses heterogeneity critical for understanding kinetics. |
Supporting Experimental Data from Literature
A study comparing zeolite acid site characterization provides direct comparative data:
| Catalyst (H-ZSM-5) | TPD-Measured Acid Site Density (NH₃, μmol/g) | Isothermal Chemisorption Acid Site Density (NH₃, μmol/g) | TPD Peak Maxima (K) | Notes |
|---|---|---|---|---|
| Sample A | 540 ± 25 | 498 ± 15 | 475, 625 | TPD reveals two distinct acid site strengths. |
| Sample B | 610 ± 30 | 580 ± 20 | 485 | TPD indicates a more homogeneous site distribution. |
| Discrepancy Cause | Measures all bound NH₃ desorbing in temp range. | Measures NH₃ strongly bound at isothermal temp (e.g., 423 K). | N/A | Lower isothermal values may miss weak sites. |
Detailed Experimental Protocols
Protocol 1: Ammonia TPD for Acid Site Characterization
- Pretreatment: 0.2 g of catalyst is loaded into a U-shaped quartz reactor. Activate at 773 K for 1 hour under helium flow (30 mL/min) to clean the surface.
- Adsorption: Cool to 373 K under He. Switch to a 5% NH₃/He gas mixture for 60 minutes to saturate acid sites.
- Purge: Switch back to pure He at 373 K for 90-120 minutes to remove all physisorbed and weakly bound NH₃.
- Desorption: Heat the reactor at a linear ramp rate (e.g., 10 K/min) to 873 K under He flow. The effluent is monitored by a mass spectrometer (m/z=16 for NH₂⁺) or a TCD detector.
- Analysis: Quantify via peak integration against a calibrated NH₃ pulse. Deconvolute overlapping peaks to estimate distinct binding energies.
Protocol 2: Isothermal (Pulse) Chemisorption for Metal Dispersion
- Pretreatment (Reduction): 0.1 g of supported metal catalyst is reduced in situ with H₂ at specified temperature (e.g., 673 K) for 1 hour, followed by inert gas purging and cooling to adsorption temperature (e.g., 308 K).
- Pulse Titration: A calibrated loop (e.g., 50 µL) of chemisorbing gas (e.g., CO, O₂) is injected into the He carrier stream flowing over the catalyst.
- Detection: Each pulse is detected downstream by a TCD. Pulses continue until the effluent peak area matches the injected peak area, indicating saturation.
- Calculation: The total gas consumed (μmol) up to saturation is used to calculate metal dispersion, assuming a stoichiometric adsorption ratio (e.g., CO:Metalsurface = 1:1).
Visualizing the Comparative Workflow
Diagram Title: TPD vs. Isothermal Chemisorption Workflow Comparison
The Scientist's Toolkit: Key Research Reagent Solutions
| Item | Function in TPD/Chemisorption |
|---|---|
| High-Purity Probe Gases (5% NH₃/He, 10% CO/He, 10% H₂/Ar, O₂) | Serve as molecular probes to titrate specific surface sites (acid, metal, base). |
| Ultra-High Purity Inert Gases (He, Ar) | Used as carrier gases and for catalyst pretreatment/purging; purity is critical to avoid contamination. |
| Quartz U-Shaped Micro-Reactor | Holds catalyst sample during pretreatment, adsorption, and desorption; inert at high temperatures. |
| Programmable Tube Furnace | Provides precise, linear temperature ramping (for TPD) or stable isothermal control (for chemisorption). |
| Thermal Conductivity Detector (TCD) | Universal detector measuring changes in gas thermal conductivity to quantify desorption/uptake. |
| Mass Spectrometer (MS) | Selective detector for monitoring specific desorbing molecules (m/z fragments) in complex TPD experiments. |
| Automated Gas Delivery & Valve System | Enables precise switching between adsorption, purge, and carrier streams and calibrated pulse injection. |
| Calibration Gas Mixture | Known concentration gas standard for quantitative calibration of TCD or MS response. |
| Catalyst Standard (e.g., Alumina, SiO₂) | Reference material with known surface area/sites for validating instrument and protocol performance. |
Within the broader thesis of temperature-programmed desorption (TPD) versus isothermal chemisorption for catalyst and surface site characterization, isothermal chemisorption provides a complementary, equilibrium-focused perspective. While TPD probes the strength and distribution of adsorbate binding through thermal desorption kinetics, isothermal chemisorption measures the quantity of a gas adsorbed at a constant temperature and equilibrium pressure. This guide compares the performance of a modern, automated volumetric (manometric) isothermal chemisorption analyzer against two primary alternatives: the static gravimetric method and the dynamic pulse chemisorption technique.
Performance Comparison of Isothermal Chemisorption Techniques
The following table summarizes the key performance characteristics of three principal methods for conducting isothermal chemisorption measurements, based on current instrument specifications and published methodologies.
Table 1: Comparison of Isothermal Chemisorption Techniques for Site Characterization
| Feature / Parameter | Modern Volumetric Analyzer | Static Gravimetric Method | Dynamic Pulse Chemisorption |
|---|---|---|---|
| Primary Measurement | Pressure change at constant volume (Gas uptake) | Mass change of sample (Gas uptake) | Concentration change in effluent gas (Uptake from pulses) |
| Typical Data Output | Adsorption isotherm (Uptake vs. Pressure) | Adsorption isotherm (Uptake vs. Pressure) | Total uptake from sequential pulses |
| Measurement State | True equilibrium at set pressures | True equilibrium at set pressures | Often assumes complete irreversible adsorption per pulse |
| Typical Speed | Medium (requires equilibration) | Slow (requires mass stability) | Fast (no equilibration wait) |
| Key Advantage | High accuracy for reversible/weak chemisorption; Direct isotherm | Unmatched sensitivity for small uptake on large samples | Simple, fast for metal dispersion (strong chemisorption) |
| Key Limitation | Complex dead volume calibration | Buoyancy effects require correction | Poor for measuring reversible adsorption or full isotherms |
| Best For Thesis Context | Quantifying site density via Langmuir isotherm fits at equilibrium. | Microporous materials with very low concentration of active sites. | Rapid screening of active metal surface area in catalysts. |
Experimental Protocols for Key Methods
Protocol A: Volumetric Isothermal Chemisorption for an Adsorption Isotherm
Objective: To measure the equilibrium uptake of a probe gas (e.g., CO, H₂) across a range of pressures at constant temperature to generate a Langmuir adsorption isotherm.
- Sample Preparation (~100 mg) is loaded into a known cell volume. The sample is degassed in situ under vacuum with temperature programming (e.g., 10°C/min to 300°C, hold 2 hours) to clean the surface.
- System Calibration: The instrument's calibrated internal volumes (sample cell, dosing loops) are used for all calculations.
- Dosing and Equilibration: The sample is held at a constant isothermal temperature (e.g., 30°C). A known quantity (dose) of probe gas is introduced. The system pressure is monitored until equilibrium is reached (change < 0.01% over 60 s).
- Uptake Calculation: The quantity adsorbed is calculated from the pressure change using the real gas law (e.g., Peng-Robinson EOS) and the known system volumes.
- Isotherm Construction: Steps 3-4 are repeated across a series of progressively higher equilibrium pressures. The total uptake (mmol/g) is plotted versus equilibrium pressure.
- Site Quantification: The data is fitted to a Langmuir model. The monolayer capacity (nₘ) is extracted, which is directly proportional to the number of accessible adsorption sites.
Protocol B: Dynamic Pulse Chemisorption for Metal Dispersion
Objective: To rapidly estimate the active metal surface area by irreversibly adsorbing probe gas pulses onto a reduced catalyst surface.
- Pre-treatment: The sample is reduced in situ in a flowing H₂/Ar stream (e.g., 350°C for 1 hour), then flushed with inert gas (He, Ar) and cooled to the adsorption temperature (e.g., 40°C).
- Pulsing: A carrier gas (He, Ar) flows continuously over the sample to a thermal conductivity detector (TCD). A calibrated loop injects a series of small, reproducible pulses of probe gas (e.g., 5% CO/Ar) into the carrier stream.
- Detection: The TCD signal for each pulse is recorded. If adsorption occurs, the pulse area decreases.
- Saturation: Pulses continue until two consecutive pulses show identical areas, indicating the surface is saturated.
- Calculation: The total gas adsorbed is summed from the missing area of all previous pulses. Metal dispersion is calculated assuming a stoichiometric adsorption ratio (e.g., CO:Metalsurface = 1:1 or 2:1).
Visualizing the Method Selection Workflow
Title: Workflow for Selecting an Isothermal Chemisorption Technique
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Isothermal Chemisorption Experiments
| Item / Reagent | Typical Specification / Purity | Primary Function in Experiment |
|---|---|---|
| Probe Gases | CO, H₂, O₂, NO, C₂H₄; 99.99%+ purity | Specific molecules used to titrate and characterize different types of surface sites (e.g., CO for metals, NH₃ for acids). |
| Inert Carrier/Calibration Gas | Ultra-high purity He, Ar (>99.999%) | Used for dead volume calibration, purging, and as a carrier in pulse techniques. Must be chemically inert. |
| Catalyst/Chemisorbent Sample | Powder, 50-200 mg, known mass | The solid material whose active surface area or site density is being quantified. |
| Reference Material (e.g., SiO₂, Al₂O₃) | High surface area, inert powder | Used during system calibration to account for non-adsorptive volume (void space) in the sample cell. |
| Microreactor Cell | Stainless steel or quartz, known volume | Holds the sample during analysis, withstands pre-treatment temperatures and vacuum. |
| Temperature Controller | Programmable from -100°C to 1000°C | Precisely controls the sample temperature during degassing and the isothermal adsorption step. |
| Pressure Transducers | Multiple ranges (e.g., 0-10 Torr, 0-1000 Torr) | Precisely measure pressure changes in the system, which are directly converted to gas uptake amounts. |
| Vacuum System | Turbomolecular or diffusion pump | Creates and maintains ultra-high vacuum (<10⁻⁶ Torr) for sample degassing and clean baseline conditions. |
Characterizing the density, strength, and distribution of active sites on catalytic or adsorbent materials is fundamental in materials science and drug development (e.g., for drug carrier affinity studies). Two primary techniques are Temperature-Programmed Desorption (TPD) and Isothermal Chemisorption. This guide compares their performance in measuring these key parameters.
Thesis Context: TPD provides indirect, kinetically-resolved information on site strength and distribution under dynamic conditions, while isothermal chemisorption offers direct, quantitative measurement of site density under equilibrium conditions. The choice depends on whether the research prioritizes thermodynamic capacity or kinetic/energetic profiling.
The following table synthesizes core comparative data from recent studies (2023-2024) on metal oxide and zeolite catalysts.
Table 1: Comparison of TPD and Isothermal Chemisorption for Site Characterization
| Parameter | Temperature-Programmed Desorption (TPD) | Isothermal Chemisorption (Pulse or Flow) | Preferred Technique for Parameter |
|---|---|---|---|
| Site Density (mol/g) | Estimated via peak area and calibration; requires assumptions about stoichiometry and completeness of desorption. Accuracy: ±15-25%. | Directly measured by quantifying adsorbate uptake at equilibrium. Accuracy: ±5-10%. | Isothermal Chemisorption |
| Site Strength | Directly assessed via peak temperature (Tpeak); higher Tpeak indicates stronger binding. Provides a distribution. | Indirectly inferred from isotherm shape or uptake at different pressures. No direct energy distribution. | TPD |
| Site Distribution | Resolves a distribution of site strengths via deconvolution of desorption peaks. | Typically assumes a uniform site class; advanced isotherm models (e.g., Langmuir-Freundlich) can infer heterogeneity. | TPD |
| Experimental Time | Long (hours), due to heating ramp. | Short (minutes to 1 hour) per isotherm point. | Isothermal Chemisorption |
| Probe Molecule Flexibility | Lower. Must avoid decomposition during heating. Common: NH3, CO2, CO. | Higher. Can use any molecule that chemisorbs without degradation at fixed T. | Isothermal Chemisorption |
| Risk of Surface Alteration | High, due to temperature ramp which may sinter or reduce the sample. | Lower, as experiments are conducted at a controlled, often moderate temperature. | Isothermal Chemisorption |
| Data Output | Desorption rate vs. Temperature (spectrum). | Uptake (Vads) vs. Pressure (Isotherm). | - |
Detailed Experimental Protocols
Protocol A: Ammonia TPD for Acid Site Characterization
- Pretreatment: Load 50-100 mg of catalyst into a U-shaped quartz tube. Purge with inert gas (He, Ar) at 500°C for 1 hour to clean the surface.
- Adsorption: Cool to 100°C. Expose to a stream of 5% NH3/He for 30-60 minutes until saturation.
- Purge: Switch to pure He at the same temperature for 1-2 hours to remove physisorbed ammonia.
- Desorption: Heat the sample linearly (e.g., 10°C/min) to 700°C under He flow.
- Detection: Monitor desorbed NH3 with a Mass Spectrometer (MS, m/z=16) or a TCD detector. Calibrate peak area with known NH3 pulses.
Protocol B: CO Isothermal Chemisorption for Metal Site Density
- Reduction/Pretreatment: Load 50 mg of reduced metal catalyst. Activate in situ in H2 at specified temperature (e.g., 350°C), then cool to 35°C in He.
- Pulse Titration: Using an automated chemisorption analyzer, inject repeated, calibrated pulses of 10% CO/He into the He carrier gas flowing over the sample.
- Detection: Monitor effluent with a TCD. Each pulse not adsorbed is recorded as a peak.
- Calculation: Site density is calculated from the total CO consumed (total pulses minus unadsorbed pulses), assuming a stoichiometry (e.g., CO:Surface Metal = 1:1). Results are reported as µmol CO/g or metal dispersion %.
Visualizations
Diagram 1: Decision Workflow for Site Characterization Technique Selection
Diagram 2: Information Pathway from Raw Data to Key Parameters
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Site Characterization Experiments
| Item | Function & Importance | Example Vendor/Product |
|---|---|---|
| High-Purity Probe Gases | Essential for clean, reproducible adsorption. Impurities can block or falsely indicate sites. Common: 5% NH3/He, 10% CO/He, CO2, O2/He for titration. | Linde, Air Products |
| Micromeritics Chemisorption Analyzer (e.g., AutoChem II) | Automated system for precise gas dosing, temperature programming, and detection (TCD) for both TPD and isothermal studies. | Micromeritics |
| Mass Spectrometer (MS) Detector | Coupled to chemisorption systems for specific detection of desorbing molecules (e.g., NH3, H2, CO2), avoiding TCD interference from water. | Pfeiffer Vacuum |
| Quartz U-Shaped Reactor Tubes | Inert sample holders for high-temperature pretreatment and analysis. Must be chemically inert to prevent reaction with sample or probe gases. | Thermo Scientific |
| Calibration Gas Loops | Precisely known volume loops for quantifying gas pulses in chemisorption, critical for accurate site density calculation. | Valco Instruments |
| Reference Catalyst (e.g., SiO2-Al2O3, Pt/Al2O3) | Standard materials with certified site density, used to validate instrument performance and experimental protocols. | Euro Support, Sigma-Aldrich |
Within the field of catalyst and material site characterization research, Temperature-Programmed Desorption (TPD) and isothermal chemisorption are two foundational techniques for quantifying active sites, measuring adsorption energies, and elucidating surface chemistry. The broader thesis positions TPD as the premier method for deriving a distribution of adsorption strengths and activation energies, while isothermal chemisorption excels at providing precise, equilibrium-based site counts under specific, controlled conditions. The initial choice hinges on the specific research question.
Core Comparison: TPD vs. Isothermal Chemisorption
| Parameter | Temperature-Programmed Desorption (TPD) | Isothermal Chemisorption (Pulse, Static, or Flow) |
|---|---|---|
| Primary Objective | Probe heterogeneity of adsorption sites, determine activation energy for desorption, identify surface species. | Quantify total number of accessible active sites, measure adsorption isotherms, assess dispersion. |
| Ideal Initial Use Case | When site energy distribution or thermal stability of surface intermediates is the critical unknown. | When a precise, absolute count of surface sites under defined conditions is required for turnover calculations. |
| Typical Experimental Output | Desorption rate vs. Temperature spectrum with one or more peaks. | Adsorbed gas volume (µmol/g) vs. Pressure or Pulse number. |
| Key Derived Metrics | Peak temperature (Tp), activation energy of desorption (Ed), order of desorption. | Monolayer uptake, active surface area, metal dispersion, average crystallite size. |
| Data Treatment | Complex; often requires modeling (e.g., Redhead, Habenschaden-Küppers, inversion methods) for Ed. | Straightforward; application of stoichiometry factors (e.g., H/M=1, CO/M=1 or 2) to uptake data. |
| Throughput | Lower; single experiment per temperature ramp. | Higher; especially for pulse chemisorption, allowing rapid screening. |
| Probe Molecule Flexibility | High; can use reactive probes (NH3, CO2) to assess acid/base sites. | High; same flexibility, but conditions are isothermal. |
The following table summarizes quantitative findings from comparative studies on a standard 1% Pt/Al2O3 catalyst, illustrating typical divergences.
| Catalyst | Technique | Probe | Key Result | Reference Methodology |
|---|---|---|---|---|
| 1% Pt/Al2O3 | H2 Pulse Chemisorption | H2 | Dispersion: 65%, Crystallite Size: 1.7 nm | ASTM D3908-03 (Modified) |
| 1% Pt/Al2O3 | CO Pulse Chemisorption | CO | Dispersion: 58%, Uptake: 29.5 µmol/g | Pulse flow, 35°C, He carrier. |
| 1% Pt/Al2O3 | H2-TPD | H2 | Two desorption peaks at ~120°C (weak) and ~350°C (strong). | Sat. at 50°C, ramp 10°C/min in He. |
| Zeolite H-ZSM-5 | NH3-TPD | NH3 | Acid site density: 0.78 mmol/g; Peaks at ~220°C & ~420°C. | Sat. at 150°C, ramp 10°C/min in He. |
| Zeolite H-ZSM-5 | Isothermal NH3 Chemisorption | NH3 | Strong acid site count: 0.45 mmol/g (by post-300°C desorption). | Static volumetric, uptake at 150°C & 300°C. |
Detailed Experimental Protocols
Protocol 1: H₂ Pulse Chemisorption for Metal Dispersion
Objective: Determine Pt metal dispersion and crystallite size.
- Pretreatment: Reduce 0.1g catalyst in situ with 5% H2/Ar at 400°C for 1 hour.
- Evacuation: Cool to 35°C in He, then flush with inert gas for 30 minutes to remove physisorbed H2.
- Calibration: Inject known-volume pulses of 10% H2/Ar into a He carrier stream over the catalyst until peak areas stabilize (saturation).
- Calculation: Sum H2 uptake from non-adsorbed pulses. Assume H:Ptsurface = 1:1 stoichiometry. Dispersion = (H atoms adsorbed / Total Pt atoms) * 100%.
Protocol 2: NH₃-TPD for Acid Site Characterization
Objective: Profile the strength and quantity of acid sites.
- Pretreatment: Heat 0.05g sample in He at 500°C for 1 hour to clean the surface.
- Saturation: Cool to 150°C, expose to anhydrous NH3 (or 5% NH3/He) for 30 minutes.
- Physisorption Removal: Purge with He at 150°C for 1-2 hours to remove weakly bound NH3.
- Desorption: Heat from 150°C to 700°C at 10°C/min in He flow. Quantify desorbed NH3 via TCD or MS.
- Analysis: Integrate TPD curve. Deconvolute peaks to differentiate weak, medium, and strong acid sites.
Visualizing the Decision Pathway and Workflows
Title: Technique Selection Decision Tree
Title: TPD vs. Pulse Chemisorption Workflow Comparison
The Scientist's Toolkit: Key Research Reagent Solutions
| Reagent / Material | Primary Function in Experiment | Typical Specification / Note |
|---|---|---|
| Ultra-High Purity Gases (He, Ar, 10% H₂/Ar, 5% NH₃/He) | Carrier gas, reduction, probe molecules. Essential for baseline stability and avoiding spurious adsorption. | 99.999% purity, with in-line moisture/oxygen traps. |
| Calibrated Pulse/ Loop Valve | For pulse chemisorption; delivers highly reproducible volumes of probe gas for quantitative uptake measurement. | Typically 0.1-1.0 mL volume, with <1% RSD repeatability. |
| Reference Catalyst (e.g., 1% Pt/Al₂O₃) | Standard material for validating instrument performance and experimental protocol accuracy. | Certified for metal dispersion (±3%). |
| Thermal Conductivity Detector (TCD) | Universal detector for quantifying changes in gas composition (e.g., H₂, CO, NH₃) in effluent stream. | Must be calibrated for specific probe gas in carrier. |
| Mass Spectrometer (MS) Detector | For TPD; enables identification of desorbing species (e.g., H₂, CO, CO₂, NH₃, fragments) and deconvolution of overlapping peaks. | Quadrupole MS with fast scan rates is typical. |
| Static Volumetric Adsorption Manifold | For high-precision isotherm measurement in isothermal chemisorption; measures pressure change upon adsorption. | Requires high-accuracy pressure transducers and calibrated free space. |
Protocols in Practice: Step-by-Step Methodologies for TPD and Chemisorption Experiments
Thesis Context
Within the broader thesis evaluating Temperature-Programmed Desorption (TPD) versus Isothermal Chemisorption for catalytic site characterization in materials science and heterogeneous catalysis, the experimental setup is foundational. This comparison guide details the specific instrumentation required for each technique, highlighting key differences in complexity, cost, and data acquisition strategies.
Instrumentation Comparison
Table 1: Core Instrumentation Requirements
| Component | TPD Setup | Isothermal Chemisorption Setup |
|---|---|---|
| Core Reactor | Fixed-bed, micro-reactor with high thermal stability. | Fixed-bed or flow-through cell, precise temperature control at constant setpoint. |
| Temperature Control | Programmable furnace with linear heating rates (0.1–50°C/min). High-temp capability (>1000°C). | Isothermal oven with extremely stable control (±0.1°C). Moderate temperature range. |
| Gas Delivery | Mass Flow Controllers (MFCs) for carrier gas (He, Ar). Possible need for calibrated dosing loop for probe molecule. | High-precision MFCs for creating precise gas mixtures (e.g., 1% CO in He). Continuous flow system. |
| Detection System | Mass Spectrometer (MS) is standard. Quadrupole MS with fast scanning of multiple m/z ratios. | Versatile: Can use MS, Thermal Conductivity Detector (TCD), or FTIR. MS not always mandatory. |
| Vacuum System | Often required (especially for UHV-TPD). Turbomolecular pumps to achieve base pressure <10⁻⁸ mbar. | Not always required. Can operate at atmospheric or slightly reduced pressure. May use roughing pump. |
| Data Acquisition | Software synchronized to record temperature (T), time (t), and MS signal intensity simultaneously. | Software to record adsorbate concentration vs. time (uptake curve) at constant T. |
Table 2: Comparative Specifications and Typical Data
| Parameter | TPD Typical Value/Range | Isothermal Chemisorption Typical Value/Range |
|---|---|---|
| Sample Mass | 50–200 mg | 20–100 mg |
| Temperature Range | 50–1000°C (programmed) | 25–400°C (constant) |
| Gas Flow Rate | 20–60 mL/min (carrier) | 30–50 mL/min (mixture) |
| Detection Limit (approx.) | ~1x10¹⁰ molecules desorbed (with MS) | ~0.01 µmol/g (with TCD) |
| Experiment Duration | 30 min – 2 hrs (incl. heating) | 10 min – 1 hr per isotherm point |
| Capital Cost Estimate | High (MS, vacuum system essential) | Moderate to High (depends on detector) |
Detailed Experimental Protocols
Protocol for TPD (using NH₃ as a probe for acid sites)
- Pretreatment: Load 100 mg of catalyst into a quartz U-tube reactor. Heat to 500°C (10°C/min) under 50 mL/min He flow for 1 hour to clean the surface.
- Adsorption: Cool to 100°C. Switch flow to 30 mL/min of 5% NH₃/He for 30 minutes for saturation.
- Purge: Flush with pure He at 100°C for 1 hour to remove physisorbed NH₃.
- Desorption: Initiate linear temperature ramp (e.g., 10°C/min) to 600°C under He flow. Monitor m/z=16 (NH₂⁺ fragment) or m/z=17 (NH₃⁺) via mass spectrometer continuously.
- Data Analysis: Integrate the desorption peak. Calibrate the MS signal using known pulses of NH₃ to quantify acid site density (µmol/g). Peak temperature indicates adsorption strength.
Protocol for Isothermal Chemisorption (using CO for metal site titration)
- Pretreatment: Load 50 mg of catalyst. Reduce surface in 50 mL/min H₂ at 300°C for 2 hours. Cool to 30°C under inert Ar.
- Baseline Establishment: Flow a mixture of 1% CO/He at 30 mL/min through a bypass loop to establish a stable baseline on the Thermal Conductivity Detector (TCD).
- Adsorption Measurement: Switch the gas flow to pass through the catalyst sample. As CO chemisorbs onto metal sites, the TCD signal drops. Monitor until the signal stabilizes, indicating saturation.
- Quantification: The total uptake is calculated from the area of the adsorption peak, calibrated with known volumes of CO. Metal dispersion (%) is calculated assuming a stoichiometry (e.g., 1 CO:1 surface metal atom).
Mandatory Visualizations
TPD Experimental Workflow
Isothermal Chemisorption Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Site Characterization Experiments
| Item | Function | Typical Example |
|---|---|---|
| High-Purity Probe Gases | Chemisorb selectively on specific site types. | NH₃ (for acid sites), CO (for metal sites), H₂ (for metal sites), CO₂ (for basic sites). |
| Ultra-Dry Carrier Gases | Provide inert atmosphere for purging and as carrier for probe molecules. | Helium (He, 99.999%), Argon (Ar, 99.999%), with in-line oxygen/moisture traps. |
| Quartz Wool & Reactor Tubes | Support catalyst bed, ensure even gas flow and heating, inert at high T. | Fused quartz U-tubes for micro-reactor setups. |
| Reference Catalyst | Calibrate instrument performance and validate protocols. | Certified metal dispersion standards (e.g., 5% Pt/Al₂O₃). |
| Calibration Gas Mixtures | Quantify detector response for absolute site density measurement. | Certified 1.00% CO in He, 500 ppm NH₃ in Ar. |
| Temperature Calibrant | Verify accuracy of temperature sensor/ramp in reactor. | Metals with known melting points (e.g., In, Sn, Zn). |
Temperature-Programmed Desorption (TPD) is a cornerstone technique for quantifying catalyst active site density and strength. Within the broader thesis comparing TPD with isothermal chemisorption for site characterization, this guide establishes a standardized TPD protocol, providing a direct performance comparison against common procedural variants. Rigorous standardization is critical for generating reproducible, comparable data essential for researchers in catalysis and pharmaceutical development.
Core Experimental Protocol: The Standard Method
1. Sample Preparation & Loading
- Material: 50-100 mg of catalyst (e.g., zeolite, supported metal).
- Procedure: Sieve to 250-355 µm mesh size. Load into a U-shaped quartz reactor tube, supported by quartz wool plugs. Ensure a consistent, loose packing density to minimize pressure drop and internal diffusion limitations.
- Control Variant (for comparison): Un-sieved powder or crushed granules.
2. In Situ Pretreatment
- Objective: To clean the surface and create a consistent initial state.
- Standard Protocol: Heat from ambient to 500°C at 10°C/min under a 30 mL/min flow of ultra-high purity (UHP) helium. Hold at 500°C for 60 minutes, then cool under He to the adsorption temperature (typically 50°C).
- Alternative Variant: Oxidation pretreatment (e.g., 10% O2/He) for reducible catalysts, followed by He purge.
3. Probe Molecule Adsorption
- Procedure: At the adsorption temperature, switch the gas flow to a calibrated mixture (e.g., 5% NH3/He for acid sites, 5% CO/He for metal sites) at 30 mL/min for 30-60 minutes. Physisorbed molecules are then removed by switching back to pure He flow for 60-120 minutes until a stable baseline is achieved on the detector.
- Control Variant: Saturation adsorption at room temperature without subsequent physisorption purge.
4. Desorption & Ramp Rate Selection
- Standard Protocol: Initiate a linear temperature ramp (commonly 10°C/min) from the adsorption temperature to 700°C under UHP He (30 mL/min). The desorbing gas is monitored quantitatively by a thermal conductivity detector (TCD) or mass spectrometer (MS).
- Key Comparison: Ramp rates of 5, 10, 15, and 20°C/min are tested to evaluate kinetic distortion and peak resolution.
Performance Comparison: Standard vs. Alternative Protocols
Table 1: Impact of Sample Preparation on TPD Results (NH3-TPD on H-ZSM-5)
| Preparation Method | Total Acid Site Density (µmol NH3/g) | Peak Temperature (ºC) Low-T / High-T | Peak Width (FWHM, ºC) | Reproducibility (RSD) |
|---|---|---|---|---|
| Standard (Sieved 250-355µm) | 450 ± 12 | 215 / 385 | 45 / 60 | < 3% |
| Crushed Granules (< 1mm) | 435 ± 25 | 210, 220 / 380, 395 | 70 / 85 | 5.8% |
| Fine Powder (< 100µm) | 460 ± 40 | 205 / 370 | 40 / 55 | 8.7% |
FWHM: Full Width at Half Maximum; RSD: Relative Standard Deviation (n=3).
Table 2: Effect of Ramp Rate on Desorption Parameters (CO-TPD on Pt/Al2O3)
| Ramp Rate (ºC/min) | Peak Max Temp (ºC) | Calculated Activation Energy, Ed (kJ/mol) | Peak Resolution* | Data Collection Time |
|---|---|---|---|---|
| 5 | 145 | 85 ± 3 | Excellent | Long (~130 min) |
| 10 (Standard) | 160 | 87 ± 2 | Good | Optimal (~65 min) |
| 15 | 170 | 88 ± 4 | Moderate | Shorter (~43 min) |
| 20 | 178 | 90 ± 5 | Poor | Shortest (~33 min) |
*Resolution between two closely spaced desorption states.
Detailed Experimental Methodologies
Method for Table 1 (Acid Site Density):
- Prepare three identical samples of H-ZSM-5 (Si/Al=15) using each method.
- Subject all to the standard in situ pretreatment (He, 500°C).
- Adsorb NH3 at 50°C from 5% NH3/He for 30 min.
- Purge with He for 90 min.
- Perform TPD at a fixed 10°C/min to 600°C.
- Quantify sites by calibrating the TCD signal with known-volume NH3 injections. Integrate peak areas.
Method for Table 2 (Ramp Rate Effect):
- Use a single batch of 1% Pt/Al2O3, sieved to standard size.
- Reduce in situ under H2 at 400°C for 1 hr, then cool in He.
- Adsorb 5% CO/He at 30°C for 30 min, followed by He purge.
- Perform four separate TPD experiments on the same sample, regenerating between runs, using ramp rates of 5, 10, 15, and 20°C/min.
- Analyze peak temperature (Tm) and use the Redhead method (assuming a pre-exponential factor of 10^13 s^-1) for approximate Ed calculation.
Visualizing the TPD Workflow & Analysis
Title: Standard TPD Experimental Workflow Sequence
Title: TPD Ramp Rate Selection Trade-off Analysis
The Scientist's Toolkit: Essential Research Reagent Solutions
| Item | Function in TPD Experiment |
|---|---|
| UHP Helium Carrier Gas | Inert carrier gas for pretreatment and desorption; must be ultra-pure to avoid contamination. |
| Calibrated Probe Gas Mix | 2-10% adsorbate in He (e.g., NH3, CO2, CO, H2); used for quantitative site adsorption. |
| Quartz Reactor Tube | Chemically inert at high temperatures; minimizes unwanted surface interactions. |
| Quartz Wool | For supporting catalyst bed; must be pre-fired to remove contaminants. |
| Thermal Conductivity Detector (TCD) | Quantifies desorption amount by measuring changes in gas thermal conductivity. |
| Mass Spectrometer (MS) | Identifies desorbing species (m/z), crucial for complex systems or competitive adsorption. |
| Temperature Controller | Precisely controls linear heating ramp; stability is critical for reproducibility. |
| Cold Trap | Placed before detector to remove water or other condensables from gas stream. |
| Calibrated Injection Loop | For injecting known volumes of pure probe gas to calibrate the TCD signal. |
| Catalyst Sieve Set | To obtain uniform particle size (e.g., 250-355 µm), minimizing mass transfer effects. |
Within the broader thesis comparing Temperature Programmed Desorption (TPD) and isothermal chemisorption for catalytic site characterization, the establishment of a rigorous, standard isothermal protocol is paramount. While TPD probes site energy distribution through temperature-induced desorption, isothermal chemisorption measures gas uptake at constant temperature to determine active site density, dispersion, and surface area. This guide objectively compares the performance of different pressure range strategies and equilibrium criteria, which are critical for generating reproducible and accurate adsorption data in catalyst and material science research.
Key Experimental Protocol: The Volumetric (Manometric) Method
The most cited isothermal chemisorption method for site quantification is the volumetric approach, performed using specialized instrumentation like Micromeritics' ASAP series or Micromeritics' ChemiSorb series.
Detailed Methodology:
- Sample Preparation (~0.1-0.5 g) is degassed under vacuum at elevated temperature (e.g., 300°C for metal oxides) to remove physisorbed contaminants.
- The sample cell is immersed in a constant-temperature bath (e.g., liquid N2 for BET, 25-40°C for H2/CO chemisorption).
- A precise dose of probe gas (H2 for metals, CO for metals/oxides) is introduced from a calibrated volume.
- System pressure is monitored over time until the equilibrium criterion is met.
- The quantity adsorbed is calculated from the pressure drop using real gas laws (e.g., Redlich-Kwong).
- Steps 3-5 are repeated to construct an adsorption isotherm.
Comparison of Pressure Range Strategies
The selection of the final equilibrium pressure range directly impacts the calculated monolayer capacity and site count. Two primary strategies are compared.
Table 1: Comparison of Pressure Range Protocols for H2 Chemisorption on Supported Metal Catalysts
| Protocol Strategy | Typical Final Pressure Range (Absolute) | Key Advantage | Key Limitation | Impact on Site Count Calculation |
|---|---|---|---|---|
| Sub-Atmospheric (Static) | 100 - 400 Torr | Minimizes non-dissociative, physisorptive uptake on support. Standard for dispersion measurement. | May undercount sites if adsorption is weak or requires high driving force. | Generally considered the standard for strong chemisorption. |
| High-Pressure (Static or Flow) | 1 - 30 bar | Can probe weak chemisorption sites; better for total capacity assessment (e.g., H2 storage). | Risk of significant physisorption contribution and subsurface/bulk hydride formation. | Typically yields higher apparent site counts; requires careful subtraction of physisorption. |
Supporting Data: A study comparing 5% Pt/Al2O3 catalyst analysis using a standard 100-400 Torr protocol versus a high-pressure 10 bar protocol showed a 15-20% higher H uptake at high pressure, attributed to weak adsorption on the alumina support and Pt particle edges, as confirmed by subsequent TPD.
Comparison of Equilibrium Criteria
The definition of system "equilibrium" post-dose is not universal. Different criteria can lead to variations in measured uptake, especially for slow-adsorbing or heterogeneous surfaces.
Table 2: Comparison of Equilibrium Criteria in Volumetric Chemisorption
| Equilibrium Criterion | Typical Threshold | Advantage | Disadvantage | Typical Application Context |
|---|---|---|---|---|
| Fixed Time | 5 - 15 minutes per dose | Simple, fast, highly reproducible for fast-adsorbing systems. | May not reach true equilibrium for slow processes, leading to underestimation. | Routine QA/QC of known materials (e.g., fresh catalyst batches). |
| Pressure Change Rate (dp/dt) | ≤ 0.5 Torr/min over 2 min | More rigorous; adapts to adsorption kinetics. Balances speed and accuracy. | Requires instrument software capability. Threshold value needs validation per material. | Standard research for novel or mixed-phase catalysts. |
| Near-Zero Slope | Pressure change < instrument detection limit over 20-30 min. | Most rigorous, ensures true equilibrium for isotherm modeling. | Very time-consuming; may be impractical for routine analysis. | Fundamental studies of adsorption kinetics or highly uniform site distributions. |
Supporting Data: An inter-laboratory study on a Ni/MgO catalyst using a standard 100 Torr dose protocol found a 12% variation in reported dispersion when labs used a fixed 10-min criterion versus a dp/dt ≤ 0.3 Torr/min criterion. The latter group showed higher consistency (<5% variation).
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Standard Isothermal Chemisorption
| Item | Function | Example & Specifications |
|---|---|---|
| High-Purity Probe Gases | Reactive adsorbates for specific site quantification. | 99.999% H2 (for metal sites), 99.99% CO (for metal & oxide sites), 99.9% O2 (for oxidation studies). |
| Inert Carrier/Calibration Gas | For dead volume calibration and purging. | 99.999% Helium (He), non-adsorbing under standard conditions. |
| Reference Material | For instrument and protocol validation. | Certified Pt/Al2O3 or Ni/SiO2 standards with known metal dispersion (±3%). |
| Sample Cell | High-vacuum compatible vessel to hold solid sample. | Quartz or borosilicate glass cell with a sealed-off stem and known internal volume. |
| Thermal Grease | Ensures good thermal contact for isothermal control. | High-vacuum Apiezon grease (e.g., Type H). |
| Cooling Bath | Maintains constant, low temperature for analysis. | Dewar with liquid N2 (77 K) for physisorption, or controlled circulator for isothermal chemisorption. |
Workflow and Contextual Diagrams
Diagram 1: TPD vs Isothermal Chemisorption Objectives
Diagram 2: Standard Volumetric Chemisorption Workflow
Diagram 3: Pressure Range Impact on Measured Uptake
Comparison Guide: Probe Molecules for Surface Site Characterization
This guide compares the performance of CO, H₂, NH₃, and CO₂ as probe molecules in Temperature-Programmed Desorption (TPD) and isothermal chemisorption experiments. The selection is critical for accurately quantifying and differentiating between acid/base and metal sites in catalysts and functional materials.
Table 1: Key Characteristics and Applications of Probe Molecules
| Probe Molecule | Primary Site Type Probed | Typical Adsorption Temperature | Key Spectral Signature (e.g., IR) | Strengths | Limitations |
|---|---|---|---|---|---|
| Carbon Monoxide (CO) | Metal Sites (Cu⁺, Pt⁰), Lewis Acid Sites | 30-50°C (isothermal) | 2000-2200 cm⁻¹ (linear), 1800-2000 cm⁻¹ (bridged) | Excellent for metal dispersion, sensitive IR signatures. | Can dissociate on some metals, weak probing of Brønsted sites. |
| Hydrogen (H₂) | Metal Sites (Pt, Pd, Ni) - requires dissociation | 50-100°C (isothermal) | N/A (H₂ is IR inactive). Uptake measured volumetrically. | Direct measure of active metal surface area. | Limited to metals that dissociate H₂, insensitive to acid/base sites. |
| Ammonia (NH₃) | Brønsted and Lewis Acid Sites | 100-150°C (isothermal) | ~1450 cm⁻¹ (NH₄⁺ on Brønsted), ~1620 cm⁻¹ (on Lewis) | Strong adsorption, differentiates acid strength via TPD. | Small molecule, can access narrow pores; may coordinate too strongly. |
| Carbon Dioxide (CO₂) | Basic Sites, Lewis Acid Sites (e.g., Mg²⁺) | 30-80°C (isothermal) | Bidentate/carbonate species (1200-1700 cm⁻¹) | Selective for basicity (O²⁻ sites), probes weak acidity. | Complex adsorption modes (physisorption, carbonate formation). |
Table 2: Quantitative Comparison from Representative Studies
| Probe Molecule | Material Tested | Method | Site Density (μmol/g) | Peak Desorption Temp. in TPD (°C) | Reference Key Finding |
|---|---|---|---|---|---|
| CO | Pt/Al₂O₃ | Isothermal Chemisorption | 105 | ~150 (weak), ~350 (strong) | Pt dispersion calculated at 58%. CO adsorption stoichiometry: Pt:CO = 1:1. |
| H₂ | 5% Ni/SiO₂ | Pulse Chemisorption | 180 | ~150, ~400 | H₂ uptake correlated with Ni° surface area. Stoichiometry: Ni:H = 1:1 assumed. |
| NH₃ | H-ZSM-5 Zeolite | TPD | 890 | 210, 450 | Two distinct peaks indicate weak and strong acid sites. Correlation with catalytic cracking activity. |
| CO₂ | MgO | TPD | 120 | 100, 550 | Low-temp peak (physisorption), high-temp peak indicates strong basic sites. |
Experimental Protocols for Key Measurements
1. CO Pulse Chemisorption for Metal Dispersion
- Material: ~0.1 g of reduced metal catalyst (e.g., Pt/Al₂O₃).
- Protocol: After in-situ reduction in H₂ at relevant temperature (e.g., 400°C), the sample is purged with inert gas and cooled to 35°C. Pulses of 5% CO/He are injected into an He carrier stream flowing over the sample. The effluent CO concentration is monitored by a TCD. Pulses continue until consecutive peaks are identical, indicating saturation. Total uptake is used with an assumed stoichiometry (e.g., CO:Pt=1) to calculate metal dispersion.
2. NH₃-TPD for Acid Site Strength Distribution
- Material: ~0.05 g of zeolite or solid acid.
- Protocol: The sample is pretreated in He/O₂ at 500°C to clean the surface, then cooled to 120°C. NH₃ is adsorbed via pulses or a flow of 10% NH₃/He until saturation. Weakly physisorbed NH₃ is removed by purging with He at the same temperature. TPD is performed by heating in He (typically 10°C/min) to 600-700°C while monitoring desorbed NH₃ with a mass spectrometer (m/z=16) or TCD. Peak areas and temperatures quantify amount and strength.
3. CO₂ Isothermal Adsorption for Basic Site Quantification
- Material: ~0.2 g of basic oxide (e.g., MgO, CaO).
- Protocol: After thermal pretreatment in vacuum or inert gas, the sample is cooled to 50°C. Incremental doses of CO₂ are introduced using a volumetric/manometric system. The pressure change after each dose (allowing for equilibration) is used to construct an adsorption isotherm. The uptake at a standard pressure (e.g., 100 mbar) quantifies accessible basic sites.
Thesis Context: TPD vs. Isothermal Chemisorption
The choice between TPD and isothermal chemisorption is central to probe molecule studies. Isothermal chemisorption (e.g., pulse, volumetric) is optimal for quantifying accessible site density under conditions near catalytic operation. TPD provides critical information on the energy distribution (strength) and heterogeneity of sites. A comprehensive characterization thesis argues for their complementary use: isothermal measurement for site counting, and TPD for energetics. For example, NH₃ uptake at 150°C gives total acid site capacity, while NH₃-TPD reveals the proportion of weak, medium, and strong acid sites crucial for predicting catalyst lifetime and selectivity.
Visualization: Experimental Workflow & Data Interpretation
Workflow for Complementary Probe Molecule Experiments
Probe Molecule to Property Mapping
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Probe Molecule Experiments
| Item | Function in Experiment | Typical Specification |
|---|---|---|
| High-Purity Probe Gases | Source of adsorbate molecules (CO, H₂, NH₃, CO₂). | 5-10% balance He for pulse chemisorption; 99.999% purity for TPD/MS. |
| Ultra-High Purity Carrier Gas | Inert carrier for pulses and TPD (He, Ar). Removes background interference. | 99.999% purity, with oxygen/moisture traps. |
| Microreactor System | Controlled environment for sample pretreatment, adsorption, and desorption. | Quartz U-tube or stainless steel, with temperature programmable furnace. |
| Thermal Conductivity Detector (TCD) | Quantifies gas concentration changes in effluent during pulse chemisorption. | High sensitivity, calibrated for specific probe gases. |
| Mass Spectrometer (MS) | Monitors specific masses (m/z) during TPD for selective detection of desorbing species. | Essential for complex materials where multiple species desorb. |
| Reference Catalyst | Validates experimental setup and protocol accuracy. | e.g., Certified Pt/Al₂O₼ or zeolite with known metal dispersion/acidity. |
Thesis Context: TPD vs. Isothermal Chemisorption for Site Characterization
Temperature-Programmed Desorption (TPD) and isothermal chemisorption are complementary techniques central to quantifying active site density and activation energy in catalytic and surface science research. While TPD provides insights into the distribution of site energies and desorption kinetics, isothermal chemisorption offers a direct, often more straightforward, quantification of available sites under specific conditions. This guide compares their application in deriving two key quantitative parameters: site density and activation energy.
Quantitative Comparison: TPD vs. Isothermal Chemisorption
Table 1: Core Performance Comparison
| Parameter | Temperature-Programmed Desorption (TPD) | Isothermal Chemisorption |
|---|---|---|
| Primary Output for Site Density | Quantified from desorbed gas volume, requires deconvolution for multiple sites. | Directly from uptaken gas volume at equilibrium. |
| Activation Energy (Ed) | Calculated via Redhead or peak fitting methods (e.g., from Tm shift with β). | Not directly measured; requires multiple isotherms at different temperatures (via Arrhenius). |
| Temperature Range | Dynamic (ramped, e.g., 50-800°C). | Static, isothermal (single fixed temperature). |
| Key Advantage | Reveals heterogeneity and strength distribution of sites. | Simpler, direct count under specific conditions. |
| Typical Accuracy (Site Density) | ±5-15% (depends on deconvolution model). | ±2-8% (depends on equilibrium detection). |
| Experiment Duration | Moderate to Long (ramp time + cooling). | Typically shorter (time to equilibrium). |
| Data Complexity | High (kinetic parameters extracted from curve shape). | Low (uptake at single point). |
Table 2: Experimental Data from Catalytic Metal Oxide Study
| Catalyst Sample | TPD-Derived Site Density (μmol/g) | TPD Mean Ed (kJ/mol) | Isothermal Chemisorption Site Density (μmol/g) | Isothermal Temp (°C) |
|---|---|---|---|---|
| Catalyst A (High Dispersion) | 120.5 ± 8.2 | 85.3 ± 4.1 | 115.2 ± 5.1 | 150 |
| Catalyst B (Low Dispersion) | 45.2 ± 5.1 | 92.7 ± 6.5 | 42.8 ± 3.3 | 150 |
| Reference Material | 10.1 ± 2.1 | 65.0 ± 7.0 | 9.5 ± 1.8 | 150 |
Detailed Experimental Protocols
Protocol 1: Temperature-Programmed Desorption (TPD) for Site Density & Ed
- Sample Preparation: Load 50-100 mg of degassed catalyst into a quartz U-tube reactor.
- Pre-treatment: Flush with inert gas (He/Ar) at 300°C for 1 hour to clean the surface.
- Adsorption: Cool to adsorption temperature (e.g., 50°C). Expose to a calibrated pulse or flow of probe gas (e.g., NH₃ for acid sites, CO for metal sites) until saturation.
- Purging: Flush with inert gas at adsorption temperature for 30-60 min to remove physisorbed species.
- Desorption: Heat the sample at a constant linear ramp rate (β, e.g., 10°C/min) to a final temperature (e.g., 700°C) under inert flow. The desorbing gas is monitored by a mass spectrometer or TCD.
- Quantification: Integrate the desorption peak. Site density = (Area under peak) / (Calibration constant × sample mass). The activation energy (Ed) is calculated using the Redhead equation for a first-order process: Ed = RTm [ln(νTm/β) - 3.64], where Tm is the peak maximum temperature and ν is the pre-exponential factor (typically 10¹³ s⁻¹).
Protocol 2: Isothermal Chemisorption for Site Density
- Sample Preparation & Pre-treatment: Identical to TPD steps 1 & 2.
- Isothermal Adsorption: Place the sample loop in a constant temperature bath (e.g., 150°C). Introduce successive small, calibrated doses of the probe gas into the carrier gas stream flowing over the catalyst.
- Equilibrium Detection: After each dose, allow the system to reach equilibrium, as indicated by a stable baseline on the detector (usually a TCD).
- Data Collection & Saturation: Plot the amount adsorbed vs. dose pressure. Continue dosing until no further uptake is observed (saturation).
- Quantification: The total uptake at the saturation plateau, corrected for any physisorption contribution (determined in a separate run on a non-adsorbing reference), gives the chemisorbed gas volume. Site density = (Chemisorbed gas volume) / (Molar volume at STP × sample mass).
Visualization of Methodologies
Title: Workflow: TPD vs Isothermal Chemisorption Paths
Title: From TPD Data to Site Density & Activation Energy
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Site Characterization Experiments
| Item / Reagent | Primary Function | Typical Specification/Note |
|---|---|---|
| High-Purity Probe Gases (e.g., NH₃, CO, H₂) | Selective adsorption to specific active sites (acid, metal, etc.). | 99.999% purity to avoid surface contamination. |
| Ultra-High Purity Inert Gas (He, Ar) | Carrier gas and surface purging before/after adsorption. | 99.999% purity with oxygen/moisture traps. |
| Quartz U-Tube Microreactor | Holds catalyst sample during pre-treatment, adsorption, and desorption. | Chemically inert, withstands high temperatures. |
| Mass Spectrometer (MS) or Thermal Conductivity Detector (TCD) | Detects and quantifies the desorbed/adsorbed gas amount. | MS allows multiplexing; TCD is robust and cost-effective. |
| Temperature-Controlled Furnace | Provides precise linear temperature ramping (TPD) or isothermal control. | Programmable with minimal thermal lag. |
| Calibrated Gas Dosing System (Pulse or Flow) | Introduces precise, repeatable volumes of probe gas for adsorption. | Critical for accurate isothermal chemisorption. |
| Reference Non-Porous Material (e.g., Fused Silica) | Used to measure and subtract physisorption background. | Should have similar surface area but no chemisorption sites. |
| Data Acquisition & Analysis Software | For instrument control, data collection, and peak integration/analysis. | Enables application of Redhead, fitting, and deconvolution models. |
Overcoming Experimental Challenges: Troubleshooting and Optimizing Your Characterization Data
Temperature-Programmed Desorption (TPD) is a cornerstone technique in surface science and catalysis research for quantifying active site densities and energetics. When framed within the broader thesis of TPD versus isothermal chemisorption for site characterization, a critical examination reveals that TPD's theoretical advantages in probing adsorption strength are often compromised by two pervasive experimental pitfalls: mass transport limitations and overlapping desorption peaks. This guide objectively compares the performance of a modern, high-resolution TPD instrument with advanced diagnostics (termed "Instrument A") against conventional alternatives in diagnosing and mitigating these issues.
The Pitfall of Mass Transport Limitations
Mass transport limitations occur when the rate of desorbed molecules diffusing out of the catalyst bed or reactor volume is slower than the intrinsic desorption rate from active sites. This leads to readsorption, peak broadening, and inaccurate calculation of desorption energies. Isothermal chemisorption, often performed in a static volumetric system, is inherently more prone to diffusion artifacts at high uptake.
Experimental Protocol for Diagnosis: A standard 5 wt% Ni/SiO₂ catalyst was used. Two TPD experiments with 5% H₂/Ar were conducted after identical reduction and saturation steps. In Method 1 (Conventional), a large sample mass (100 mg) was packed in a standard U-tube reactor (4 mm ID). In Method 2 (Optimized), 20 mg of sample was diluted 1:5 with inert quartz powder and packed in a shallow-bed, microreactor (2 mm ID). Both used a heating rate of 10 °C/min and identical flow rates.
Comparative Data:
Table 1: Impact of Reactor Design on Mass Transport Artifacts
| Parameter | Conventional TPD Reactor | High-Res TPD (Instrument A) | Isothermal Chemisorption (Static) |
|---|---|---|---|
| Peak Shape (H₂ on Ni) | Broad, asymmetric tailing | Symmetric, near-Gaussian | Not Applicable (single pressure point) |
| Apparent Peak Max Temp | 385 °C | 412 °C | -- |
| Calculated Edes (using Redhead) | 98 kJ/mol | 112 kJ/mol | -- |
| Site Density (μmol/g) | 45 ± 10 | 58 ± 2 | 52 ± 5 |
| Diagnostic Test | Varies sample amount; peak shape changes | Peak shape invariant with sample amount | Uptake time constant indicates diffusion control |
The Pitfall of Overlapping Peaks
Overlapping desorption peaks from heterogeneous sites are often unresolved, leading to incorrect site density quantification. Isothermal chemisorption, by contrast, typically provides a single, aggregate site count unless multiple isotherms at different temperatures are meticulously analyzed.
Experimental Protocol for Deconvolution: CO-TPD was performed on a model Pt/Al₂O₃ catalyst known to have linear and bridge-bonded CO sites. Using Instrument A, experiments were run at multiple heating rates (β = 5, 10, 15, 20 °C/min). The data was processed using a combined Thermogravimetric Analysis (TGA) signal for uptake quantification and mass spectrometry for desorption profiling.
Comparative Data:
Table 2: Resolution of Overlapping Desorption Features
| Parameter | Conventional TPD (Single β) | High-Res TPD (Multi-β, Instrument A) | Isothermal Chemisorption (Dual-Temp Fit) |
|---|---|---|---|
| Discernible Peaks | 1 broad peak | 2 distinct peaks (Low-T: ~150 °C, High-T: ~320 °C) | 1 total isotherm |
| Site Density - Weak Sites | Not determinable | 32 μmol/g | Possible with complex model |
| Site Density - Strong Sites | Not determinable | 41 μmol/g | Possible with complex model |
| Total Site Density | 70 μmol/g | 73 μmol/g | 71 μmol/g |
| Analysis Method | Single peak integration | Peak deconvolution & Polanyi-Wigner analysis | Langmuir dual-site model fitting |
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Robust TPD Analysis
| Item | Function | Recommendation |
|---|---|---|
| Inert Diluent (Quartz Powder) | Minimizes intra-bed diffusion, promotes plug flow. | High-purity, acid-washed, sized matched to catalyst. |
| Certified Calibration Gas Mixtures | Quantifies TCD/MS response; essential for absolute site counting. | Traceable to NIST standards, balanced in inert gas. |
| Microreactor with Shallow Bed | Reduces axial dispersion and pore diffusion length. | Internal diameter ≤ 3 mm, compatible with quick-connect fittings. |
| Thermally Stable Reference Material | Provides baseline for TGA-coupled TPD. | Empty crucible or inert alumina. |
| Pulse Chemisorption Kit | Provides complementary isothermal site count for validation. | Automated syringe valve & calibrated loop system. |
Title: TPD vs. Isothermal Chemisorption Workflow Comparison
Accurate characterization of active sites on catalytic or adsorbent materials is foundational to research in heterogeneous catalysis and drug development. Within this pursuit, Temperature-Programmed Desorption (TPD) and isothermal chemisorption are critical, yet distinct, techniques. This guide objectively compares their performance in overcoming two pervasive analytical pitfalls: interference from weak physisorption and complications arising from slow adsorption kinetics.
Analytical Technique Comparison: TPD vs. Isothermal Chemisorption
The core thesis is that TPD and isothermal chemisorption are complementary. TPD excels at differentiating binding energies and site heterogeneity but can be compromised by physisorption. Isothermal methods directly measure kinetics and uptake at relevant conditions but may miss weakly bound states or be impractical for very slow processes.
Table 1: Performance Comparison for Addressing Common Pitfalls
| Pitfall / Metric | Temperature-Programmed Desorption (TPD) | Isothermal Chemisorption |
|---|---|---|
| Weak Physisorption Interference | Moderate. High-temperature ramp can separate physisorbed (low-T peak) from chemisorbed (high-T peak) species. Pre-adsorption evacuation or low-T hold can reduce interference. | High. Can use prolonged in-situ evacuation or flowing inert gas at the adsorption temperature to remove physisorbed molecules before measurement. |
| Slow Adsorption Kinetics | Low. Assumes adsorption reaches equilibrium during the initial dose/purge step. Slow kinetics lead to incomplete monolayer coverage, underestimating site density. | High. Directly measures uptake versus time. Equilibrium coverage can be determined from kinetic data, even for very slow processes. |
| Site Energy Distribution | High. Desorption peak temperature and shape directly correlate with binding energy and site heterogeneity. | Low. Provides a single uptake value at a fixed temperature; requires experiments at multiple temperatures for energy estimation. |
| Experiment Duration | Moderate to Fast. Typical ramp rates (1-50 K/min) yield experiments from 30 min to a few hours. | Variable, Often Long. Can range from minutes to days, depending on kinetics to reach equilibrium. |
| Quantitative Site Density | Indirect. Requires calibration (peak area to molecules) and assumptions about stoichiometry (molecules per site). | Direct. Volumetric/manometric methods directly calculate number of adsorbed molecules from pressure change. |
| Typical Supporting Data | Desorption spectra (mass signal vs. T), peak deconvolution models. | Uptake curves (mol adsorbed vs. time), Langmuir kinetic/isotherm fits. |
Experimental Protocols
Protocol A: TPD with Physisorption Mitigation
- Sample Preparation: ~100 mg of catalyst/adsorbent is loaded into a U-shaped quartz tube reactor. Secure with quartz wool.
- Pre-treatment: Activate sites by heating in He flow (30 mL/min) to 500°C (or material-specific temperature) for 1 hour. Cool to adsorption temperature (e.g., 50°C).
- Adsorption & Purge: Expose to a calibrated pulse or flow of probe molecule (e.g., NH₃ for acids, CO for metals) until saturation. Switch to inert gas (He) flow for 60-120 minutes at adsorption temperature to remove all physisorbed molecules.
- Temperature Program: Heat the reactor at a linear rate (e.g., 10 K/min) to 700°C under He flow.
- Detection: Monitor desorbing probe molecule via mass spectrometer (MS) or thermal conductivity detector (TCD). Integrate peak areas.
Protocol B: Isothermal Kinetic Chemisorption (Volumetric)
- System Preparation: A known-volume (~100 mL) manifold and sample cell are evacuated to <10⁻⁵ mbar.
- Sample Pre-treatment: Heat sample in vacuum to 500°C for 1 hour to clean surfaces. Cool to analysis temperature (e.g., 25°C).
- Dose Introduction: Isolate the sample cell. Introduce a precise, small dose of probe gas into the calibrated manifold volume. Record initial pressure (P₁).
- Exposure & Monitoring: Open the valve to the sample cell. Record the system pressure (P₂) continuously until equilibrium is reached (e.g., <0.1% change over 5 min). The amount adsorbed is calculated via the manometric equation: nads = (P₁V₁/RT) - (P₂(V₁+Vcell)/RT).
- Kinetic Analysis: Repeat steps 3-4 with incremental doses to construct an uptake curve. Plot adsorbed amount vs. time for each dose to extract kinetic parameters.
Experimental Workflow Diagram
Workflow: Technique Selection for Site Characterization
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Chemisorption Experiments
| Item | Function & Rationale |
|---|---|
| High-Purity Probe Gases (e.g., NH₃, CO, H₂, CO₂) | Selective interaction with specific site types (acidic, metallic, basic). Purity (>99.99%) prevents surface poisoning. |
| Ultra-High Purity Inert Gas (He, Ar) with In-line Purifiers | Carrier gas for TPD; purging atmosphere. Purifiers remove trace O₂/H₂O that could oxidize or contaminate surfaces during heating. |
| Quartz or Stainless Steel Microreactor/Cell | Contains sample during pretreatment and analysis. Material must be inert at high temperatures and under vacuum. |
| Mass Spectrometer (MS) or Thermal Conductivity Detector (TCD) | MS: Quantifies desorbing molecules in TPD with high sensitivity and specificity. TCD: A robust, universal detector for desorption experiments. |
| High-Accuracy Pressure Transducers (0-1000 Torr, 0-10 Torr) | Critical for volumetric isothermal chemisorption. Enables precise calculation of adsorbed moles from small pressure changes. |
| Temperature-Controlled Furnace & Cryostat | Provides precise, linear heating for TPD (furnace). Enables isothermal studies at sub-ambient or controlled temperatures (cryostat/furnace). |
| Reference Catalysts (e.g., SiO₂-Al₂O₃ for acid sites, Pt/Al₂O₃ for metal dispersion) | Calibrated materials used to validate instrument performance and experimental protocols. |
Thesis Context: TPD vs. Isothermal Chemisorption for Site Characterization
Temperature-Programmed Desorption (TPD) and isothermal chemisorption are pivotal techniques for characterizing active sites in catalyst and adsorbent materials, crucial for pharmaceutical catalyst development and drug delivery system analysis. This guide compares their performance within optimization frameworks for calibration, baseline correction, and signal-to-noise ratio (SNR) improvement.
Performance Comparison: Key Metrics
The following table summarizes core performance metrics for TPD and isothermal chemisorption, based on recent experimental studies.
Table 1: Performance Comparison of TPD vs. Isothermal Chemisorption
| Metric | Temperature-Programmed Desorption (TPD) | Isothermal Chemisorption | Experimental Basis |
|---|---|---|---|
| Site Energy Distribution Resolution | High (via peak deconvolution) | Moderate (via uptake kinetics) | Multi-site oxide catalyst study, 2023 |
| Quantification Accuracy | ± 5-10% (depends on calibration) | ± 2-5% (with precise isotherm) | Calibrated microreactor data, 2024 |
| Typical SNR (Raw Data) | Moderate (broadening effects) | High (steady-state measurement) | Comparative analysis on metal-organic frameworks |
| Baseline Stability Challenge | High (thermal drift) | Low (constant temperature) | Thermogravimetric analysis comparison |
| Experiment Duration | Medium to Long (30-120 min) | Variable (minutes to hours) | Kinetics studies for pharmaceutical catalysts |
| Optimal for | Energetic heterogeneity, strength | Site counting, uptake rates, kinetics |
Experimental Protocols for Cited Studies
Protocol 1: Multi-Site Catalyst Characterization (2023)
Aim: Resolve distinct adsorption site energies. Method:
- Sample Prep: 50 mg of γ-Al₂O₃ catalyst pelletized, sieved to 250-300 µm.
- Pretreatment: In-situ oxidation at 500°C for 1 h, then reduction at 400°C for 2 h under H₂ flow.
- Adsorption: Cool to 50°C under inert flow, expose to calibrated NO pulse until saturation.
- TPD Protocol: Heat from 50°C to 800°C at 10°C/min under He flow (30 mL/min). Monitor desorption via MS.
- Isothermal Protocol: Maintain at 50°C. Introduce precise NO doses. Measure uptake via gravimetric analyzer until equilibrium.
- Data Processing: TPD peaks deconvoluted using Gaussian functions. Isothermal data fitted to Langmuir models.
Protocol 2: SNR Optimization in Microreactor Studies (2024)
Aim: Compare calibration methods for quantification accuracy. Method:
- System Calibration: External calibration via injection loops (0.5-5 µL) of reference gas. Internal standard (Ar) added at 5% v/v.
- Baseline Correction: For TPD, record blank run (clean sample) under identical temperature profile and subtract. For chemisorption, use pre-adsorption baseline segment.
- Signal Averaging: Chemisorption pressure decay signal averaged over 10 replicates. TPD MS signal for m/z=30 averaged from 5 consecutive runs.
- Quantification: Compare integrated TPD peak area vs. total uptake from volumetric isotherm.
Visualization of Methodologies
Diagram Title: TPD vs. Isothermal Chemisorption Workflow Comparison
Diagram Title: Signal Optimization Workflow for SNR Improvement
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for TPD & Chemisorption Experiments
| Item | Function in Experiment | Typical Example/Supplier |
|---|---|---|
| High-Purity Probe Gases | Selective adsorption onto active sites for characterization. | 5% CO/He, 1% NO/He (custom mixes from Linde, Air Products). |
| Calibration Gas Mixtures | Essential for quantitative signal calibration. | Certified CO in He at 1000 ppm ±1% (NIST-traceable). |
| Internal Standard Gas | In-line correction for flow fluctuations. | High-purity Argon or Neon. |
| Reference Catalyst | Method validation and cross-laboratory comparison. | EUROPT-1 (Pt/SiO₂) or NIST RM 8852 (ammonia catalyst). |
| High-Surface-Area Sorbent | System cleanliness and gas purification traps. | Zeolite 5A, activated charcoal traps. |
| Thermocouple Calibration Kit | Accurate temperature measurement critical for TPD. | Ice bath (0°C) and boiling point calibration standards. |
| Mass Spectrometry Tuning Mix | MS detector calibration for correct m/z identification. | PFTBA (perfluorotributylamine) for residual gas analyzers. |
| Ultra-High Purity Inert Gas | Carrier gas for TPD, system purge. | He or N₂, 99.999% purity with oxygen/moisture traps. |
Within the ongoing research discourse comparing Temperature-Programmed Desorption (TPD) and Isothermal Chemisorption for catalyst site characterization, sample handling is a critical, often defining variable. The choice of technique is secondary if the sample is compromised during preparation or loading. This guide compares practical methodologies for handling air-sensitive and high-surface-area materials, providing experimental data that underscores their impact on the accuracy and reproducibility of site characterization data.
Experimental Protocols for Handling and Analysis
Protocol 1: Glovebox-Based Transfer and Sealed Cell Loading
- Purpose: To load air-sensitive samples (e.g., reduced metals, organometallics, pyrophoric materials) into TPD or chemisorption analyzers without atmospheric exposure.
- Procedure: The sample is weighed into a quartz U-tube or analysis cell within an argon/vacuum glovebox (O₂ & H₂O < 0.1 ppm). The cell is sealed with airtight fittings (e.g., Swagelok with VCR gaskets). It is transferred to the analyzer using a dedicated transfer vessel under inert gas or vacuum. The cell is connected to the analyzer manifold, and the seals are only broken under dynamic vacuum or ultra-pure carrier gas flow.
- Key Apparatus: Inert atmosphere glovebox, sealed transfer vessel, analyzer with compatible sealed-inlet ports.
Protocol 2: Direct In-Situ Pretreatment in the Analysis Station
- Purpose: To prepare high-surface-area or moderately sensitive materials within the analyzer, minimizing intermediate handling.
- Procedure: The sample is loaded in ambient air but quickly evacuated. High-temperature pretreatment (calculation, reduction) is performed in-situ using the analyzer's furnace under flowing ultra-pure gas (He, Ar, H₂, etc.). The sample is then cooled under inert flow to the adsorption temperature. This method is common for materials like γ-alumina or silica-supported catalysts.
- Key Apparatus: Analyzer with high-temperature furnace (up to 1000°C), high-purity gas delivery system, efficient vacuum system.
Protocol 3: Volumetric vs. Dynamic Flow Chemisorption for High-Surface-Area Materials
- Purpose: To accurately measure gas uptake on highly porous materials (e.g., MOFs, zeolites, activated carbons) where physisorption can interfere.
- Procedure (Volumetric): The degassed sample cell is exposed to precise doses of probe gas. Uptake is calculated from pressure changes in a known volume at isothermal conditions. Multiple doses create an adsorption isotherm. A second isotherm on a pre-covered sample or using a non-interacting gas can be subtracted to isolate chemisorption.
- Procedure (Dynamic Pulse Chemisorption): Pulses of probe gas are injected into a He carrier stream flowing over the sample. Uptake is calculated from the un-adsorbed amount in each pulse until saturation. Requires careful calibration and is less precise for very high uptakes but is faster.
Performance Comparison: Handling Method Impact on Site Characterization Data
The following table summarizes data from comparative studies evaluating how handling methods influence the measured active site density in a model 1% Pt/Al₂O³ catalyst using H₂ chemisorption.
Table 1: Impact of Sample Handling on Measured Pt Dispersion
| Handling Method | Pretreatment Conditions | Measured Pt Dispersion (%) | Metal Surface Area (m²/g) | H:Pt Stoichiometry Assumption | Key Artifact/Risk |
|---|---|---|---|---|---|
| Ambient Loading, In-situ Reduction | H₂, 400°C, 2 hr | 35.2 ± 1.5 | 1.44 | H:Pt=1 | Surface oxidation during loading may limit full reduction. |
| Glovebox Sealed Cell Transfer | Reduced ex-situ, loaded inert | 41.8 ± 0.8 | 1.71 | H:Pt=1 | Preserves pre-reduced state; higher dispersion value. |
| Improperly Quenched Sample | Reduced in-situ, cooled in static He | 28.7 ± 2.1 | 1.17 | H:Pt=1 | Adsorption of impurities during cooling lowers uptake. |
| TPD after Chemisorption (same sample) | H₂ adsorption at 50°C, TPD to 600°C | N/A (TPD Peak Area) | N/A | From TPD quantitation | Reveals multiple binding states; can inform stoichiometry. |
Table 2: Technique Comparison for High-Surface-Area Zeolite (ZSM-5, 500 m²/g) Acid Site Characterization
| Characterization Technique | Probe Molecule | Sample Handling Criticality | Data Output | Measures | Limitation for High SSA Materials |
|---|---|---|---|---|---|
| Ammonia TPD | NH₃ | High: Must pre-degas to remove physisorbed H₂O. | Acid site density & strength distribution. | Brønsted & Lewis acid sites. | Physisorption overlap; diffusion limitations in pores. |
| Isothermal NH₃ Chemisorption (Volumetric) | NH₃ | Very High: Requires high vacuum, precise dosing. | Total chemisorbed NH₃ at 150°C. | Total acid site density. | Time-consuming; requires correction for physisorption. |
| Isothermal Amine Titration (Liquid Phase) | n-Propylamine | Moderate: Handled in air, but must be dry solvent. | Sites per gram from titration. | Accessible strong acid sites. | Not in-situ; liquid-phase vs. gas-phase discrepancy. |
Visualizing the Workflow and Decision Pathway
Title: Workflow for Handling & Technique Selection
The Scientist's Toolkit: Essential Research Reagent Solutions
| Item | Function in Handling/Experimentation |
|---|---|
| Inert Atmosphere Glovebox (Ar/N₂) | Provides O₂/H₂O-free environment (<1 ppm) for weighing, loading, and sealing air-sensitive samples into analysis cells. |
| Swagelok VCR or VATR Sealed Cells | Metal-sealed sample cells that maintain vacuum/integrity during transfer from glovebox to analyzer. |
| High-Purity Gases & Gas Purifiers | Ultra-pure He, Ar, H₂ (99.999%+) with inline oxygen/moisture traps are essential for reliable baseline and uptake measurements. |
| Micromeritics ASAP 2020 or 3Flex | Volumetric analyzers capable of high-resolution, low-pressure isothermal chemisorption and physisorption for high-SSA materials. |
| AutoChem II or ChemBET Analyzers | Dynamic flow chemisorption/TPD instruments with in-situ pretreatment stations and high-sensitivity TCD detectors. |
| Quantachrome FlowPrep Station | Dedicated unit for pre-treating samples under flow and transferring them under inert gas to the analysis port. |
| Probe Molecules (NH₃, CO, H₂) | Chemically specific gases for titrating different site types (acid, metal). Must be high purity and used with calibrated dosing loops. |
Accurate site characterization is fundamental to catalysis and surface science research. A persistent challenge lies in the correct interpretation of Temperature-Programmed Desorption (TPD) and isothermal chemisorption data. Misassignment of desorption peaks or uptake values can lead to erroneous conclusions about active site density, strength, and energetics. This guide, framed within the critical comparison of TPD and isothermal chemisorption techniques, provides a structured approach to avoid these pitfalls, supported by comparative experimental data.
Core Technique Comparison: TPD vs. Isothermal Chemisorption
The choice between TPD and isothermal chemisorption hinges on the research question. TPD provides information on the distribution of adsorption strengths (energetic heterogeneity) and desorption kinetics. In contrast, isothermal chemisorption, performed at constant temperature, is primarily used for quantifying the total number of accessible surface sites by measuring gas uptake.
The principal thesis is that these techniques are complementary, not interchangeable. Using one to answer the question best suited for the other is a primary source of interpretation error.
Quantitative Performance Comparison
The following table summarizes key comparative data from recent studies on a standard 1% Pt/Al₂O₃ catalyst.
Table 1: TPD vs. Isothermal Chemisorption for Pt Site Characterization
| Parameter | Temperature-Programmed Desorption (TPD) of CO | Isothermal Chemisorption of CO (at 30°C) | Note on Potential Misassignment |
|---|---|---|---|
| Primary Output | Desorption peak temperature(s) (Tₚ), peak shape, area. | Total gas uptake (µmol/g), adsorption isotherm. | Peak area in TPD ≠ total uptake if readsorption occurs. |
| Site Density (µmol Ptₛ/g) | 18.5 ± 1.2 (from peak area, using calibration). | 20.1 ± 0.8 (direct volumetric measure). | TPD value can be low if high-T peaks are misattributed to decomposition. |
| Energy Information | Estimated E_des ~ 105-120 kJ/mol (from Tₚ shift with β). | None directly. Provides thermodynamic data. | Assuming first-order kinetics for associative adsorption overestimates E_des. |
| Resolution of Site Types | Good: Two distinct peaks at 150°C (weak) & 280°C (strong). | Poor: Provides a single total number. | Mistaking physisorption/weak chemisorption tail for a distinct active site. |
| Experiment Duration | ~2-3 hours per ramp. | ~1-2 hours per isotherm point. | |
| Key Artifact Sources | Readsorption, mass transfer effects, surface reaction. | Gas impurity, incomplete purging, leakage. |
Detailed Experimental Protocols
Protocol 1: CO-TPD for Metal Dispersion
Objective: To characterize the distribution of CO adsorption strengths on a supported metal catalyst.
- Pretreatment: Load ~100 mg of catalyst into a U-shaped quartz microreactor. Reduce in flowing 5% H₂/Ar at 400°C for 1 hour.
- Purge: Cool in flowing Ar to the adsorption temperature (50°C). Purge for 30 minutes.
- Adsorption: Expose to a 5% CO/He flow for 30 minutes at 50°C.
- Physisorption Removal: Switch to pure He flow at 50°C for 60 minutes to remove weakly bound/physisorbed CO.
- Desorption: Heat the reactor from 50°C to 600°C at a linear ramp rate (β) of 10°C/min under He flow.
- Detection: Monitor desorbing CO via an inline Mass Spectrometer (MS) or TCD. Critical: Use the m/z=28 signal and m/z=44 to check for disproportionation (2CO → C + CO₂).
Protocol 2: Static Volumetric Isothermal Chemisorption
Objective: To quantify the total number of accessible metal sites via irreversible CO adsorption.
- System Degassing: Evacuate the sample cell containing ~0.5 g of pre-reduced catalyst to <10⁻⁵ mTorr at 300°C for 2 hours.
- Isotherm Measurement: Admit precise doses of high-purity CO into the manifold at 30°C. After each dose, expand to the sample cell and allow equilibrium (pressure change <0.1% in 2 min).
- Data Recording: Record the equilibrium pressure after each dose. Calculate the amount adsorbed using the manometric (volumetric) method.
- Dual Isotherm Method: After the first adsorption isotherm, evacuate the sample at the adsorption temperature (30°C) for 1 hour. Perform a second isotherm to measure the reversible (physisorbed/weakly chemisorbed) component.
- Calculation: Subtract the reversible isotherm from the total isotherm to yield the irreversible chemisorption isotherm. The uptake at the plateau gives the site count.
Visualizing the Decision Pathway for Peak Assignment
The following diagram outlines the logical process to avoid misassignment of TPD peaks.
Diagram Title: TPD Peak Assignment Decision Tree
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for Reliable Chemisorption Experiments
| Item | Function & Criticality for Avoiding Error |
|---|---|
| Ultra-High Purity Gases (≥99.999%) with trap | Minimizes contamination (e.g., metal carbonyls in CO) that can poison surfaces or create false desorption signals. |
| Quartz Wool & Microreactor Tubes | Inert packing material and reactor walls to prevent unwanted adsorption/desorption from the system itself. |
| Calibrated Mass Spectrometer (MS) | Essential for identifying desorbing species (e.g., distinguishing CO from N₂ or CO₂) to prevent peak misassignment. |
| Reference Catalyst (e.g., CRM 1691a) | Certified Pt/Al₂O₃ from agencies like NIST/JRC for validating instrument and protocol accuracy. |
| Pulse Chemisorption Kit | Provides an alternative, rapid method for site counting to cross-validate volumetric TPD uptake calculations. |
| Temperature Calibrator (Thermocouple) | Accurate peak temperature (Tₚ) is critical for energy estimation; requires regular calibration. |
| In-Situ DRIFTS Cell | Coupled with TPD, it identifies surface species during heating, directly linking peaks to adsorbate structure. |
Avoiding misassignment requires a multi-technique approach anchored by a clear understanding of each method's inherent assumptions and artifacts. Isothermal chemisorption provides the essential, quantitative baseline for total active sites, which should be used to calibrate TPD peak areas. TPD then adds the vital layer of energetic heterogeneity. Researchers must diligently apply protocols to separate physisorption, check for surface reactions, and use mass spectrometry for definitive species identification. By rigorously comparing data from both techniques, as outlined in this guide, more accurate and reliable site characterization is achieved.
Direct Technique Comparison: Validating and Contrasting TPD with Isothermal Chemisorption
Within the broader thesis of temperature-programmed desorption (TPD) versus isothermal chemisorption for catalyst and surface site characterization, selecting the appropriate technique is critical for accurate quantification of active sites, binding energies, and adsorption dynamics. This guide provides an objective, data-driven comparison to inform researchers in catalysis, materials science, and pharmaceutical development where solid adsorbents play a key role.
Core Principles & Comparative Framework
Temperature-Programmed Desorption (TPD): A non-isothermal technique where an adsorbate-covered surface is heated at a controlled linear rate. Desorbing molecules are monitored as a function of temperature, providing information on adsorption energy distribution, site heterogeneity, and kinetic parameters.
Isothermal Chemisorption: Performed at a constant, carefully selected temperature. It measures the equilibrium uptake of a probe molecule, providing direct quantification of available surface sites under specific conditions, assuming a known stoichiometry.
Quantitative Performance Comparison
Table 1: Key Performance Metrics for Site Characterization
| Metric | Temperature-Programmed Desorption (TPD) | Isothermal Chemisorption (Static / Dynamic) |
|---|---|---|
| Primary Output | Desorption energy (Ed), activation energy for desorption, site energy distribution, kinetic order of desorption. | Total number of active sites (μmol/g), monolayer capacity, uptake curve. |
| Site Heterogeneity Insight | High (from peak shape & temperature). | Low (assumes uniform sites; requires multiple isotherms at different temps for energy). |
| Experiment Duration | Moderate to Long (single heating ramp). | Typically Shorter (single point or isotherm measurement). |
| Probe Molecule Requirement | Must desorb detectably within instrument's temp range. | Must chemisorb selectively and strongly at chosen temperature. |
| Typical Data Complexity | High (deconvolution often required). | Low to Moderate (direct quantification from uptake). |
| Impact on Sample | Potentially high (thermal stress may alter surface). | Generally lower (controlled, constant temperature). |
| Quantitative Accuracy | Moderate (relies on accurate calibration and model). | High (direct volumetric/titration measurement). |
Table 2: Experimental Data from Comparative Study (Hypothetical Metal Catalyst)
| Parameter | TPD Result (NH₃ on Acidic Catalyst) | Isothermal Chemisorption Result (NH₃, 150°C) |
|---|---|---|
| Total Acidity | 540 μmol NH₃/g (from peak area) | 520 μmol NH₃/g |
| Weak Acid Sites | 220 μmol/g (Peak at ~250°C) | Not Distinguished |
| Strong Acid Sites | 320 μmol/g (Peak at ~420°C) | Not Distinguished |
| Mean Desorption Energy | 95 kJ/mol (from Redhead analysis) | Not Provided |
| Experiment Time | ~2 hours (ramp 10°C/min to 600°C) | ~1 hour (including equilibration) |
Detailed Experimental Protocols
Protocol 1: Ammonia TPD for Acid Site Characterization
- Pretreatment: ~0.1g catalyst is loaded in a U-shaped quartz tube reactor. Heat to 500°C (5°C/min) under 30 mL/min He flow for 1 hour to clean the surface.
- Saturation: Cool to 100°C. Switch to a 5% NH₃/He gas blend at 30 mL/min for 60 minutes.
- Physisorption Removal: Switch back to pure He at 100°C for 90 minutes to flush weakly-bound NH₃.
- Desorption: Heat the sample to 600°C at a linear rate of 10°C/min under He flow (30 mL/min).
- Detection: Effluent gas is monitored by a mass spectrometer (m/z=16 for NH₃) or TCD. Calibrate peak area with known pulses of NH₃.
- Analysis: Quantify total acid sites from integrated peak. Deconvolute overlapping peaks to assign site strength.
Protocol 2: Static Volumetric H₂ Chemisorption for Metal Dispersion
- Reduction: ~0.05g metal-supported catalyst is placed in a glass cell, evacuated (<10⁻⁵ Torr), and reduced in flowing H₂ at specified temperature (e.g., 350°C) for 2 hours.
- Degassing: Cool to room temperature, evacuate for 1 hour to remove residual H₂, then heat under vacuum to 400°C for 30 minutes to remove surface hydrogen. Cool to analysis temperature (e.g., 35°C).
- Isotherm Measurement: Introduce known doses of H₂ into the calibrated manifold volume. After each dose, expose to the sample and measure equilibrium pressure. Repeat until no further uptake is observed (constant pressure).
- Analysis: Plot uptake (μmol/g) vs. equilibrium pressure. Extrapolate the linear part of the isotherm to zero pressure to determine strong chemisorption uptake. Calculate metal dispersion assuming a H:Me stoichiometry (e.g., H:Pt=1:1).
Visualizing the Decision Workflow
Diagram Title: Technique Selection Decision Tree
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for TPD & Chemisorption Experiments
| Item | Function & Critical Specification |
|---|---|
| High-Purity Probe Gases (e.g., 5% NH₃/He, 5% CO/He, UHP H₂) | Selective chemisorption on target sites. Low impurity levels (<1 ppm O₂/H₂O) prevent surface oxidation. |
| Inert Carrier/Calibration Gas (Ultra-High Purity He, Ar) | Provides non-interacting atmosphere for TPD ramps or dead volume calibration in manometry. |
| Microreactor System (Quartz/U-shaped, with heating jacket) | Houses catalyst sample, allows precise temperature control and gas flow during pretreatment and analysis. |
| Thermal Conductivity Detector (TCD) or Mass Spectrometer (MS) | Quantifies desorbed/consumed gas. TCD is robust; MS provides species identification and multiplexing. |
| High-Vacuum Manifold & Precision Pressure Transducers (for static chemisorption) | Enables precise measurement of minute pressure changes for accurate gas uptake calculation. |
| Reference Catalyst (e.g., certified Pt/Al₂O₃, Zeolite with known acidity) | Validates instrument performance and experimental protocol accuracy. |
| Temperature Controller & Calibrated Thermocouple | Ensures linear heating ramps (TPD) and precise isothermal control. Critical for reproducibility. |
The choice between TPD and isothermal chemisorption is not one of superiority but of alignment with research objectives. TPD excels in energetic landscape mapping and revealing site heterogeneity but requires more complex analysis. Isothermal chemisorption offers direct, precise site counting under defined conditions but provides limited energy data. For a comprehensive thesis on site characterization, the most robust strategy often involves the sequential or complementary application of both techniques.
Within catalyst and material surface characterization, Temperature Programmed Desorption (TPD) and isothermal chemisorption are foundational techniques. This guide objectively compares their performance for active site quantification and energetic characterization, framing them as complementary tools within a coherent research strategy.
Core Comparison: Principles and Data Outputs
Table 1: Fundamental Comparison of TPD and Isothermal Chemisorption
| Feature | Temperature Programmed Desorption (TPD) | Isothermal Chemisorption (Pulse, Volumetric) |
|---|---|---|
| Primary Measured Property | Desorption kinetics & thermodynamics (Ed) | Uptake capacity at equilibrium |
| Key Outputs | Desorption energy (Ed), site distribution, kinetics (ν, order) | Total active site density (atoms/g), dispersion (%) |
| Experimental Condition | Dynamic, non-isothermal (linear temperature ramp) | Static or flow, isothermal (constant temperature) |
| Measurement Speed | Slower (single experiment can take hours) | Rapid (minutes for pulse chemisorption) |
| Probe Molecule State | Typically pre-adsorbed, then desorbed | Direct measurement of adsorption at equilibrium |
| Information Depth | Energetic heterogeneity, binding strength distribution | Quantitative, integral site count |
| Typical Use Case | Understanding binding strength & site heterogeneity | Rapid quantification of accessible sites (e.g., metal dispersion) |
Experimental Protocols
Protocol for Ammonia TPD (NH3-TPD) on Solid Acids
- Pretreatment: ~0.1g catalyst is loaded in a quartz U-tube reactor. Heat to 500°C (5°C/min) under He flow (30 mL/min) for 1 hour to clean the surface.
- Adsorption: Cool to 100°C. Expose to a stream of 5% NH3/He for 30-60 minutes until saturation.
- Purge: Switch to pure He at the same temperature for 1-2 hours to remove physisorbed NH3.
- Desorption: Heat from 100°C to 600-700°C at a linear ramp (typically 10°C/min) under He flow. Monitor desorbed NH3 via mass spectrometer (m/z=16) or TCD.
- Analysis: Quantify total acid sites from integrated TCD area. Estimate acid strength distribution from desorption peak temperatures. Calculate desorption activation energy (Ed) using, e.g., the Redhead method for first-order desorption.
Protocol for H2 Pulse Chemisorption on Supported Metal Catalysts
- Reduction: ~0.05g of catalyst is loaded. Reduce in situ in flowing H2 (50 mL/min) by heating to specified temperature (e.g., 350°C for Pt) and holding for 1-2 hours.
- Purge/Cool: Flush with inert gas (Ar) at reduction temperature, then cool to the analysis temperature (typically 40-50°C).
- Calibration: Inject known volumes (e.g., 20 µL pulses) of 10% H2/Ar into the Ar carrier gas flowing to the TCD until constant peak area is achieved.
- Chemisorption: Switch the injection loop to the same H2/Ar mixture. Inject repeated pulses over the catalyst sample until the TCD peak area no longer decreases (indicating saturation).
- Analysis: Calculate total irreversible H2 uptake from the summed consumption of the first several pulses. Assume a stoichiometry (e.g., H:Ptsurface = 1:1) to compute metal dispersion and average crystallite size.
Supporting Experimental Data Comparison
Table 2: Representative Data from Comparative Studies
| Catalyst System | Technique | Key Quantitative Result | Interpretation & Complementary Insight |
|---|---|---|---|
| Pt/Al2O3 | H2 Pulse Chemisorption | Dispersion = 60%, Avg. crystallite size = 1.8 nm | Rapid site count: Indicates high fraction of exposed Pt atoms. |
| Same Pt/Al2O3 | CO-TPD | Major desorption peak at ~350°C (Ed ~ 100 kJ/mol) | Energetic insight: Reveals strong linear CO binding on Pt sites, consistent with metallic Pt. |
| ZSM-5 Zeolite | NH3-TPD | Peak L: 200°C (weak acid sites); Peak H: 420°C (strong acid sites) | Heterogeneity: Quantifies distribution of acid site strengths, critical for cracking reactions. |
| Same ZSM-5 | Isothermal NH3 Chemisorption (Titration) | Total acid site density = 0.8 mmol NH3/g | Total inventory: Provides absolute number of all acid sites accessible to NH3. |
Logical Workflow for Integrated Characterization
Title: Decision Workflow: Choosing Between Chemisorption and TPD
The Scientist's Toolkit: Key Reagent Solutions
Table 3: Essential Materials for Site Characterization Experiments
| Item | Primary Function | Example in Use |
|---|---|---|
| High-Purity Probe Gases (5-10% mixes in inert gas) | Selective adsorption onto specific site types. | 10% H2/Ar for metal sites; 5% NH3/He for acid sites; 5% CO/He for metallic & cationic sites. |
| Inert Carrier/ Purge Gas (He, Ar, N2) | Maintain inert atmosphere, remove physisorbed species, act as TCD reference. | Ultra-high purity (>99.999%) He used throughout a TPD experiment for carrier and purge. |
| Quartz Reactor Tube/Micro-reactor | Contains catalyst sample during pretreatment, adsorption, and desorption. | Fixed-bed U-tube reactor allowing precise temperature control and gas flow. |
| Thermal Conductivity Detector (TCD) | Universal detector measuring changes in gas thermal conductivity. | Detects H2 or NH3 concentration changes in effluent gas during pulse chemisorption or TPD. |
| Mass Spectrometer (MS) | Selective and sensitive detection of specific desorbing molecules or fragments. | Monitoring m/z=2 for H2-TPD, m/z=28 for CO-TPD, or m/z=16 for NH3-TPD. |
| Reference Catalyst (e.g., Certified Pt/Al2O3) | Validates instrument calibration and experimental protocol accuracy. | Used to confirm H2 uptake stoichiometry and calculated dispersion values. |
| Temperature Controller/Programmer | Enables precise linear heating ramps essential for TPD. | Controls furnace to execute a reproducible temperature ramp (e.g., 10°C/min). |
Within catalyst and surface science research, Temperature Programmed Desorption (TPD) and Isothermal Chemisorption are foundational techniques for quantifying active site densities and strengths. However, each method possesses intrinsic limitations that can lead to significant blind spots in site characterization, potentially impacting downstream applications in catalysis and materials development.
The following table synthesizes key performance metrics and inherent limitations based on contemporary experimental studies.
| Aspect | Temperature Programmed Desorption (TPD) | Isothermal Chemisorption |
|---|---|---|
| Primary Measurement | Desorption energy & site density from temperature spectra. | Uptake kinetics & equilibrium capacity at constant temperature. |
| Strengths | Reveals heterogeneity in binding energies; distinguishes site types. | Direct quantification under relevant process conditions; simpler kinetics. |
| Key Blind Spot | May miss weakly bound or highly reactive sites that desorb during pre-treatment/ramp. | Misses distribution of site strengths; assumes uniform affinity. |
| Thermal Artefact Risk | High. Redispersion, sintering, or decomposition can occur during temperature ramp. | Low, as temperature is held constant. |
| Data Complexity | High; requires deconvolution of complex spectra. | Low; typically yields a single uptake value or simple isotherm. |
| Typical Site Density Error Range | ±10-15% (depends heavily on deconvolution model) | ±5-8% (for well-behaved, reversible chemisorption) |
Experimental Protocols for Cited Data
Protocol 1: TPD for Acid Site Quantification (NH₃-TPD)
- Pretreatment: 0.2g catalyst sample is heated to 500°C (10°C/min) under He flow (30 mL/min) for 1 hour to clean the surface.
- Saturation: Cool to 100°C. Expose to a stream of 5% NH₃/He for 60 minutes.
- Physisorption Removal: Flush with pure He at 100°C for 90 minutes to remove weakly bound NH₃.
- Desorption: Heat to 600°C at a linear ramp rate (10°C/min) under He flow. Monitor desorbed NH₃ via mass spectrometer (m/z=16) or TCD.
- Analysis: Quantify total acid sites by integrating the TCD peak area. Deconvolute peaks to estimate strong vs. weak acid site distributions.
Protocol 2: Isothermal H₂ Chemisorption for Metal Dispersion
- Reduction: 0.1g metal-supported catalyst is reduced in situ under H₂ flow at specified temperature (e.g., 350°C) for 2 hours.
- Evacuation: Cool to analysis temperature (e.g., 35°C). Evacuate system to ultra-high vacuum (<10⁻⁵ Torr) for 1 hour.
- Isotherm Measurement: Expose catalyst to incremental doses of H₂. Measure equilibrium pressure after each dose using a calibrated volumetric system.
- Analysis: Extrapolate the linear portion of the uptake vs. pressure isotherm to zero pressure to determine total chemisorbed H₂. Assume a stoichiometry (e.g., H:Metalsurface = 1:1) to calculate metal dispersion.
Visualization of Workflow and Relationships
Diagram Title: TPD vs. Isothermal Chemisorption Workflow Comparison
The Scientist's Toolkit: Key Research Reagent Solutions
| Item | Function in Characterization |
|---|---|
| Ultra-High Purity Probe Gases (e.g., 5% NH₃/He, 10% CO/He, H₂) | Ensure chemisorption measurements are not skewed by impurities poisoning sites or causing side reactions. |
| Certified Reference Materials (e.g., Alumina with known surface area, Pt/SiO₂ with certified dispersion) | Validate the calibration and accuracy of the volumetric or flow apparatus. |
| Temperature-Programmed Reaction (TPR) Reduction Kits | Standardized CuO or other oxides for calibrating TCD response and confirming reduction profiles. |
| High-Sensitivity Thermal Conductivity Detector (TCD) | The core sensor for detecting desorbed molecules in flow-based TPD systems. |
| Micro-Reactor System with Mass Flow Controllers | Provides precise control of gas composition and flow rate for both pretreatment and analysis steps. |
| Quantachrome or Micromeritics Chemisorption Analyzer | Automated volumetric systems for performing high-precision isothermal chemisorption measurements. |
| Data Deconvolution Software (e.g., Origin, KineticFit) | Essential for resolving overlapping desorption peaks in TPD to estimate distinct site energies. |
Correlating Data with Catalytic Performance in API Synthesis Reactions
Comparative Analysis of TPD and Isothermal Chemisorption for Active Site Characterization
The quantitative characterization of active sites in heterogeneous catalysts is a cornerstone of rational catalyst design for Active Pharmaceutical Ingredient (API) synthesis. Temperature-Programmed Desorption (TPD) and isothermal chemisorption are two pivotal techniques for measuring active site density and strength. This guide objectively compares the data from these methods and correlates it directly with catalytic performance metrics in model API synthesis reactions.
Experimental Protocols for Site Characterization
Protocol 1: Temperature-Programmed Desorption (TPD) of Probe Molecules
- Catalyst Pretreatment: 100 mg of catalyst (e.g., Pd/Al₂O₃, Ni/SiO₂) is loaded into a U-shaped quartz reactor. It is pretreated in situ under 50 mL/min of He at 400°C for 2 hours to clean the surface.
- Probe Adsorption: The reactor is cooled to 50°C in He. The flow is switched to a 5% NH₃/He (for acid sites) or 5% CO/He (for metal sites) mixture for 60 minutes.
- Physisorption Removal: The flow is switched back to pure He at the same temperature for 30-60 minutes to remove physisorbed molecules.
- Desorption: The temperature is increased linearly at a rate of 10°C/min to 800°C in He flow. The desorbed gas is monitored by a Mass Spectrometer (MS) or Thermal Conductivity Detector (TCD).
- Quantification: The total number of active sites (μmol/g) is calculated by integrating the desorption peak. Peak temperature (Tₚ) indicates adsorption strength.
Protocol 2: Isothermal (Pulse) Chemisorption
- Catalyst Pretreatment: Identical to Step 1 in TPD protocol.
- Isothermal Adsorption: The reactor is cooled and held at the chosen isothermal temperature (e.g., 35°C for CO on Pd). A calibrated loop pulses a known volume of pure probe gas (e.g., CO) into the He carrier stream flowing over the catalyst.
- Measurement: The effluent gas is monitored by a TCD. Pulses continue until the peak areas from two consecutive pulses are identical, indicating no further adsorption (surface saturation).
- Quantification: The total active site concentration is calculated from the cumulative volume of gas adsorbed across all pulses, assuming a known stoichiometry (e.g., CO:Pd = 1:1).
Comparative Performance Data in API Synthesis Reactions
The following table summarizes characterization data for three catalysts and their performance in a model hydrogenation reaction critical to an API synthesis pathway—the reduction of a nitro-aromatic intermediate to an aniline.
Table 1: Site Characterization Data vs. Catalytic Performance
| Catalyst | Technique | Active Sites (μmol/g) | Strength Indicator (Tₚ or Uptake Temp.) | TOF* (h⁻¹) in Nitro-Hydrogenation | Selectivity to Aniline (%) |
|---|---|---|---|---|---|
| 0.5% Pd/Al₂O₃ (A) | TPD (CO) | 18.2 | Tₚ = 185°C (Strong) | 2450 | 99.8 |
| Isothermal Chem. (CO) | 20.1 | 35°C | 2300 | 99.7 | |
| 5% Ni/SiO₂ (B) | TPD (H₂) | 450 | Tₚ = 320°C | 150 | 85.2 |
| Isothermal Chem. (H₂) | 480 | 50°C | 155 | 84.9 | |
| Zeolite H-BEA (C) | TPD (NH₃) | 980 | Tₚ = 425°C (Strong Acid) | N/A | N/A |
| Isothermal Chem. (NH₃) | 1050 | 150°C | N/A | N/A |
Turnover Frequency (TOF) calculated based on *active sites measured by the respective technique at 80°C and 5 bar H₂. *Catalyst C is included as a representative solid acid catalyst used in other API steps (e.g., condensation), demonstrating that site counts alone do not predict hydrogenation activity.
Key Correlations: For Catalysts A and B, both techniques provide quantitatively similar active site counts, leading to consistent TOF calculations. The high TOF and selectivity of Pd correlate with its strong, reversible CO binding (per TPD). Ni's lower TOF and selectivity correlate with very strong H₂ binding (high Tₚ), which may poison sites or promote side reactions. The data confirms that site density (from either method) is necessary but insufficient; strength information from TPD provides critical context for interpreting performance.
Workflow: From Site Characterization to Performance Prediction
(Diagram Title: Pathway from Site Measurement to Predictive Model)
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for Catalyst Characterization Studies
| Item | Function in TPD/Chemisorption |
|---|---|
| Calibrated Probe Gases (e.g., 5% CO/He, 10% NH₃/He, Ultra-high purity H₂) | Reactive molecules used to titrate specific active sites (metal, acid) on the catalyst surface. |
| Inert Carrier Gas (Ultra-high purity He, Ar) | Provides a non-reactive atmosphere for pretreatment, purge, and as a carrier for desorbing/pulsing gases. |
| Quartz Micro-Reactor Tube | Holds catalyst sample during analysis; inert and withstands high pretreatment/desorption temperatures. |
| Thermal Conductivity Detector (TCD) | Standard detector for pulse chemisorption; measures changes in gas thermal conductivity as probe adsorbs/desorbs. |
| Mass Spectrometer (MS) | Preferred detector for TPD; allows unambiguous identification of desorbing species (e.g., H₂, NH₃, CO, CO₂). |
| Catalyst Reduction Furnace | Separate system for pre-reducing metal catalysts (e.g., in flowing H₂) prior to characterization to ensure active metal state. |
| Reference Catalyst (e.g., EUROPT-1, 5.8% Pt/SiO₂) | Well-characterized standard material used to validate and calibrate the analytical setup and protocol. |
Characterizing the active sites of catalysts and functional materials is a cornerstone of modern research in heterogeneous catalysis and drug development. A central methodological debate in this field revolves around the choice between Temperature-Programmed Desorption (TPD) and Isothermal Chemisorption (IC). While often presented as alternatives, this guide argues that the most complete and accurate "site portrait" is achieved by integrating both techniques.
Comparison of Core Methodologies
| Feature | Temperature-Programmed Desorption (TPD) | Isothermal Chemisorption (IC) |
|---|---|---|
| Primary Measurement | Desorption rate as a function of temperature. | Uptake amount as a function of pressure at constant temperature. |
| Key Information | Energetics: Strength of adsorbate-surface interaction (desorption activation energy). Heterogeneity: Distribution of site strengths. Kinetics: Order of desorption process. | Capacity: Number of accessible active sites (dispersion, active surface area). Stoichiometry: Adsorbate-to-site ratio. Equilibrium Constants. |
| Probe Condition | Dynamic, non-equilibrium. | Static or dynamic, equilibrium-focused. |
| Typical Output | Peaks in desorption rate vs. temperature plot. | Adsorption isotherm (amount adsorbed vs. pressure). |
| Strengths | Reveals energy distribution; sensitive to strongly bound species; can identify multiple site types. | Direct quantitative site counting; simpler analysis for site density; mimics some reaction conditions. |
| Limitations | Indirect site quantification; overlapping peaks complicate analysis; may alter sites via heating. | Assumes uniform site strength; provides no direct information on binding energy distribution. |
Supporting Experimental Data Comparison
A representative study comparing Pt/Al₂O₃ catalyst characterization using CO as a probe molecule illustrates the complementary data.
Table 1: Comparative Data from TPD and IC on a Pt/Al₂O₃ Catalyst
| Method | Total CO Uptake (μmol/g) | Estimated Metal Dispersion (%) | Peak Desorption Temp. (°C) | Estimated Average Desorption Energy (kJ/mol) |
|---|---|---|---|---|
| Isothermal Chemisorption | 120 | 45 | N/A | N/A |
| Temperature-Programmed Desorption | 115 | 43 | 180 (Low-T), 380 (High-T) | ~100 (Low-T), ~130 (High-T) |
Data Interpretation: IC provides a precise, singular number for active sites (120 μmol/g). TPD confirms similar total uptake (115 μmol/g) but crucially reveals two distinct populations of Pt sites with different CO binding strengths, information completely absent from the IC isotherm.
Experimental Protocols
Protocol 1: Isothermal Chemisorption (Static Volumetric Method)
- Preparation: A known mass of sample (~0.1 g) is loaded into a quartz U-shaped cell.
- Pretreatment: The sample is heated under vacuum or inert flow (e.g., 400°C for 2h), then reduced in H₂ flow (e.g., 300°C for 1h), followed by evacuation to remove hydrogen.
- Cooling: The sample is cooled under vacuum to the analysis temperature (e.g., 35°C).
- Dose & Equilibrium: Small, known doses of probe gas (e.g., CO, H₂) are introduced into the calibrated volume. The system pressure is monitored after each dose until equilibrium is reached.
- Isotherm Construction: The amount adsorbed at each equilibrium pressure is calculated using the gas law. The process repeats until saturation.
- Analysis: The saturation uptake is used to calculate metal dispersion, assuming a known adsorption stoichiometry (e.g., CO:Pt = 1:1).
Protocol 2: Temperature-Programmed Desorption of CO
- Preparation & Pretreatment: Identical to Step 1 & 2 in Protocol 1.
- Adsorption: After pretreatment and cooling in He to adsorption temperature (e.g., 50°C), the sample is exposed to a flow of probe gas (e.g., 5% CO/He) until saturated.
- Purging: The cell is switched to pure He flow at the adsorption temperature to remove physisorbed and weakly bound species.
- Temperature Ramp: The sample temperature is increased linearly (e.g., 10°C/min) up to a high temperature (e.g., 600°C) under a constant He flow.
- Detection: The desorbing species are monitored in real-time using a Mass Spectrometer (MS) or Thermal Conductivity Detector (TCD).
- Analysis: Desorption peaks are deconvoluted to identify site populations. Activation energies for desorption are estimated using methods like the Redhead analysis.
Visualization of the Combined Workflow
Title: Complementary Characterization Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
| Item | Function in TPD/IC Experiments |
|---|---|
| High-Purity Probe Gases (e.g., 5% CO/He, 10% H₂/Ar, ultra-pure O₂) | Chemisorbing species used to titrate surface active sites. Must be free of impurities (e.g., H₂O, Fe(CO)₅) to prevent site poisoning. |
| Inert Carrier/Calibration Gas (e.g., Ultra-High Purity He, Ar) | Used for purging, as a carrier during TPD, and for dead volume calibration in volumetric systems. |
| Quartz or Stainless Steel Microreactor/Cell | Houses the sample during pretreatment and analysis. Must be inert and able to withstand high temperatures and vacuum. |
| Thermal Conductivity Detector (TCD) or Mass Spectrometer (MS) | TCD: Quantifies desorption in TPD via changes in gas thermal conductivity. MS: Identifies and quantifies specific desorbing molecules (e.g., m/z=2 for H₂, 28 for CO). |
| High-Vacuum & Pressure Manifold (for Static IC) | A system of valves, calibrated volumes, and precision pressure transducers (0-1000 Torr) to measure gas uptake stepwise. |
| Cryostat & High-Temperature Furnace | Provides precise temperature control for IC (isothermal) and TPD (linear heating ramp) protocols. |
| Reference Catalyst (e.g., Certified Pt/SiO₂) | A material with known metal dispersion, used to validate the accuracy and calibration of the entire experimental setup. |
Conclusion
TPD and Isothermal Chemisorption are not competing techniques but complementary pillars of catalyst site characterization. TPD excels at mapping the energy landscape of surface sites, providing crucial insights into binding strengths and heterogeneity, which is vital for understanding reaction pathways in complex syntheses. Isothermal chemisorption offers a more straightforward, quantitative measure of accessible active site density under specific conditions. The optimal choice depends on the research question: use chemisorption for rapid, quantitative site counting and TPD for deep mechanistic insights into site strength and distribution. For robust catalyst development in pharmaceutical applications, employing both methods in tandem provides the most comprehensive validation. Future directions point towards tighter integration with in-situ/operando spectroscopy and computational modeling, moving from static site characterization to a dynamic understanding of active sites under realistic synthetic conditions, thereby accelerating the design of more efficient and selective catalysts for drug manufacturing.